Abstract
Aims
Arbuscular mycorrhizal (AM) fungi establish root symbioses that can improve acquisition of nutrients such as nitrogen (N) from soils, leading to improvements in host plant growth. Previously, we showed that applying AM fungal necromass to the rhizosphere could stimulate plant growth. Here we investigated the interactions between AM fungi (living or dead) and ammonia oxidizers (AO), a key group of soil prokaryotes involved in soil N cycling, to understand the effects of these interactions on plant N levels. We were interested in whether the necromass contained any significant biological nitrification inhibitory (BNI) activity that could account for the previously suggested antagonism between AM fungi and AO.
Methods
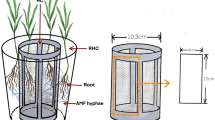
We set up a compartmented pot experiment with Andropogon gerardii as a host plant, which included rhizosphere (amended with living and/or dead AM fungal biomass) and a root-free zone. The latter contained a mesh bag with 15N-labelled plant litter.
Results
Inoculation with living AM fungus enhanced plant biomass and nutrient acquisition, including increased transfer of 15N from the mesh bags to the plants. Additionally, AO bacteria but not archaea were significantly suppressed in the mesh bags. In contrast, AM fungal necromass had minimal effect on plant biomass and nutrient acquisition. Surprisingly, it stimulated the growth of both AO bacteria and archaea in the rhizosphere of non-mycorrhizal plants.
Conclusions
Based on the above, we found no support for AM fungi suppressing the AO through an elusive BNI production. Rather, it seems that the main mechanism of AM fungal-AO interaction is substrate competition.







Similar content being viewed by others
Data availability
Original data of the research used for data analysis are available in Supplementary Information (supplementary file3). The paired sequences (after initial quality filter but before rarefaction and 18S amplicon splitting to protistan and fungal sequences) generated in this study were deposited in the Sequence Read Archive under accession number PRJNA943160.
References
Bernhardt ES, Blaszczak JR, Ficken CD et al (2017) Control points in ecosystems: moving beyond the hot spot hot moment concept. Ecosystems 20:665–682. https://doi.org/10.1007/s10021-016-0103-y
Booth MS, Stark JM, Rastetter E (2005) Controls on nitrogen cycling in terrestrial ecosystems: a synthetic analysis of literature data. Ecol Monogr 75:139–157. https://doi.org/10.1890/04-0988
Brundrett MC (2009) Mycorrhizal associations and other means of nutrition of vascular plants: understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant Soil 320:37–77. https://doi.org/10.1007/s11104-008-9877-9
Brundrett MC, Tedersoo L (2018) Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol 4:1108–1115. https://doi.org/10.1111/nph.14976
Bücking H, Kafle A (2015) Role of arbuscular mycorrhizal fungi in the nitrogen uptake of plants: current knowledge and research gaps. Agronomy 5:587–612. https://doi.org/10.3390/agronomy5040587
Bukovská P, Bonkowski M, Konvalinková T et al (2018) Utilization of organic nitrogen by arbuscular mycorrhizal fungi-is there a specific role for protists and ammonia oxidizers? Mycorrhiza 28:269–283. https://doi.org/10.1007/s00572-018-0825-0
Bukovská P, Rozmoš M, Kotianová M et al (2021) Arbuscular mycorrhiza mediates efficient recycling from soil to plants of nitrogen bound in chitin. Front Microbiol 12:574060. https://doi.org/10.3389/fmicb.2021.574060
Byrnes RC, Nùñez J, Arenas L et al (2017) Biological nitrification inhibition by Brachiaria grasses mitigates soil nitrous oxide emissions from bovine urine patches. Soil Biol Biochem 107:156–163. https://doi.org/10.1016/j.soilbio.2016.12.029
Chandrasekaran M (2022) Arbuscular mycorrhizal fungi mediated enhanced biomass, root morphological traits and nutrient uptake under drought stress: a meta-analysis. J Fungi 8:660. https://doi.org/10.3390/jof8070660
Chaudhary VB, Rúa MA, Antoninka A et al (2016) MycoDB, a global database of plant response to mycorrhizal fungi. Sci Data 3:1–10. https://doi.org/10.1038/sdata.2016.28
Coca-Salazar A, Richaume A, Florio A, Carnol M (2021) Response of ammonia-oxidizing bacteria and archaea abundance and activity to land use changes in agricultural systems of the Central Andes. Eur J Soil Biol 102:103263. https://doi.org/10.1016/j.ejsobi.2020.103263
Coskun D, Britto DT, Shi W, Kronzucker HJ (2017) Nitrogen transformations in modern agriculture and the role of biological nitrification inhibition. Nat Plants 3:1–10. https://doi.org/10.1038/nplants.2017.74
Daims H, Lebedeva EV, Pjevac P et al (2015) Complete nitrification by nitrospira bacteria. Nature 528:504–509. https://doi.org/10.1038/nature16461
Dudáš M, Pjevac P, Kotianová M et al (2022) Arbuscular mycorrhiza and nitrification: disentangling processes and players by using synthetic nitrification inhibitors. Appl Environ Microbiol 88:e01369-e1422. https://doi.org/10.1128/aem.01369-22
Faghihinia M, Jansa J, Halverson LJ, Staddon PL (2023) Hyphosphere microbiome of arbuscular mycorrhizal fungi: a realm of unknowns. Biol Fertil Soils 59:17–34. https://doi.org/10.1007/s00374-022-01683-4
Garcia K, Doidy J, Zimmermann SD et al (2016) Take a trip through the plant and fungal transportome of mycorrhiza. Trends Plant Sci 21:937–950. https://doi.org/10.1016/j.tplants.2016.07.010
Geisen S, Koller R, Hünninghaus M et al (2016) The soil food web revisited: diverse and widespread mycophagous soil protists. Soil Biol Biochem 94:10–18. https://doi.org/10.1016/j.soilbio.2015.11.010
Gryndler M, Šmilauer P, Püschel D et al (2018) Appropriate nonmycorrhizal controls in arbuscular mycorrhiza research: a microbiome perspective. Mycorrhiza 28:435–450. https://doi.org/10.1007/S00572-018-0844-x
Hawkins H-J (2000) Uptake and transport of organic and inorganic nitrogen by arbuscular mycorrhizal fungi. Plant Soil 226:275–285. https://doi.org/10.1023/A:1026500810385
Hoeksema JD, Chaudhary VB, Gehring CA et al (2010) A meta-analysis of context-dependency in plant response to inoculation with mycorrhizal fungi. Ecol Lett 13:394–407. https://doi.org/10.1111/j.1461-0248.2009.01430.x
Jansa J, Forczek ST, Rozmoš M et al (2019) Arbuscular mycorrhiza and soil organic nitrogen: network of players and interactions. Chem Biol Technol Agric 6:1–10. https://doi.org/10.1186/s40538-019-0147-2
Jansa J, Šmilauer P, Borovička J et al (2020) Dead Rhizophagus irregularis biomass mysteriously stimulates plant growth. Mycorrhiza 30:63–77. https://doi.org/10.1007/s00572-020-00937-z
Jiang F, Zhang L, Zhou J et al (2021) Arbuscular mycorrhizal fungi enhance mineralisation of organic phosphorus by carrying bacteria along their extraradical hyphae. New Phytol 230:304–315. https://doi.org/10.1111/nph.17081
Johansen A, Jakobsen I, Jensen ES (1993) Hyphal transport by a vesicular-arbuscular mycorrhizal fungus of N applied to the soil as ammonium or nitrate. Biol Fertil Soils 16:66–70. https://doi.org/10.1007/bf00336518
Lata JC, Le Roux X, Koffi KF et al (2022) The causes of the selection of biological nitrification inhibition (BNI) in relation to ecosystem functioning and a research agenda to explore them. Biol Fertil Soils 58:207–224. https://doi.org/10.1007/s00374-022-01630-3
Leininger S, Urich T, Schloter M et al (2006) Archaea predominate among ammonia-oxidizing prokaryotes in soils. Nature 442:806–809. https://doi.org/10.1038/nature04983. (4427104)
Li Z, Zeng Z, Tian D et al (2020) Global patterns and controlling factors of soil nitrification rate. Glob Chang Biol 7:4147–4157. https://doi.org/10.1111/gcb.15119
Liu B, Han F, Ning P et al (2022) Root traits and soil nutrient and carbon availability drive soil microbial diversity and composition in a northern temperate forest. Plant Soil 479:281–299. https://doi.org/10.1007/s11104-022-05516-z
Lu Y, Zhang X, Ma M et al (2022) Syringic acid from rice as a biological nitrification and urease inhibitor and its synergism with 1,9-decanediol. Biol Fertil Soils: 1–13. https://doi.org/10.1007/s00374-021-01584-y
Mendes R, Garbeva P, Raaijmakers JM (2013) The rhizosphere microbiome: significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms. FEMS Microbiol Rev 37:634–663. https://doi.org/10.1111/1574-6976.12028
Ngwene B, Gabriel E, George E (2013) Influence of different mineral nitrogen sources (NO−3-N vs. NH+4-N) on arbuscular mycorrhiza development and N transfer in a Glomus intraradices–cowpea symbiosis. Mycorrhiza 23:107–117. https://doi.org/10.1007/s00572-012-0453-z
Nuccio EE, Blazewicz SJ, Lafler M et al (2022) HT-SIP: a semi-automated stable isotope probing pipeline identifies cross-kingdom interactions in the hyphosphere of arbuscular mycorrhizal fungi. Microbiome 101:1–20. https://doi.org/10.1186/s40168-022-01391-z
Ohno T, Zibilske LM (1991) Determination of low concentrations of phosphorus in soil extracts using malachite green. Soil Sci Soc Am J 55:892–895. https://doi.org/10.2136/sssaj1991.03615995005500030046x
Prosser JI, Nicol GW (2012) Archaeal and bacterial ammonia-oxidisers in soil: the quest for niche specialisation and differentiation. Trends Microbiol 20:523–531. https://doi.org/10.1016/j.tim.2012.08.001
Qu Q, Zhang Z, Peijnenburg WJGM et al (2020) Rhizosphere microbiome assembly and its impact on plant growth. J Agric Food Chem 68:5024–5038. https://doi.org/10.1021/acs.jafc.0c00073
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Řezáčová V, Gryndler M, Bukovská P et al (2016) Molecular community analysis of arbuscular mycorrhizal fungi—Contributions of PCR primer and host plant selectivity to the detected community profiles. Pedobiologia (Jena) 4:179–187. https://doi.org/10.1016/j.pedobi.2016.04.002
Rotthauwe JH, Witzel KP, Liesack W (1997) The ammonia monooxygenase structural gene amoa as a functional marker: Molecular fine-scale analysis of natural ammonia-oxidizing populations. Appl Environ Microbiol 63:4704–4712. https://doi.org/10.1128/aem.63.12.4704-4712.1997
Rozmoš M, Bukovská P, Hršelová H et al (2022) Organic nitrogen utilisation by an arbuscular mycorrhizal fungus is mediated by specific soil bacteria and a protist. ISME J 16:676–685. https://doi.org/10.1038/s41396-021-01112-8
Schauss K, Focks A, Leininger S et al (2009) Dynamics and functional relevance of ammonia-oxidizing archaea in two agricultural soils. Environ Microbiol 11:446–456. https://doi.org/10.1111/j.1462-2920.2008.01783.x
Smith SE, Smith FA, Jakobsen I (2004) Functional diversity in arbuscular mycorrhizal (AM) symbioses: the contribution of the mycorrhizal P uptake pathway is not correlated with mycorrhizal responses in growth or total P uptake. New Phytol 162:511–524. https://doi.org/10.1111/j.1469-8137.2004.01039.x
Spang A, Poehlein A, Offre P et al (2012) The genome of the ammonia-oxidizing Candidatus Nitrososphaera gargensis: insights into metabolic versatility and environmental adaptations. Environ Microbiol 14:3122–3145. https://doi.org/10.1111/j.1462-2920.2012.02893.x
Subbarao GV, Nakahara K, Hurtado MP et al (2009) Evidence for biological nitrification inhibition in Brachiaria pastures. Proc Natl Acad Sci USA 106:17302–17307. https://doi.org/10.1073/pnas.0903694106
Sun R, Myrold DD, Wang D et al (2019) AOA and AOB communities respond differently to changes of soil pH under long-term fertilization. Soil Ecol Lett 1:126–135. https://doi.org/10.1007/s42832-019-0016-8
Tao R, Wakelin SA, Liang Y, Chu G (2017) Response of ammonia-oxidizing archaea and bacteria in calcareous soil to mineral and organic fertilizer application and their relative contribution to nitrification. Soil Biol Biochem 114:20–30. https://doi.org/10.1016/j.soilbio.2017.06.027
Tatsumi C, Taniguchi T, Du S et al (2020) Soil nitrogen cycling is determined by the competition between mycorrhiza and ammonia-oxidizing prokaryotes. Ecology 101:e02963. https://doi.org/10.1002/ecy.2963
Thonar C, Erb A, Jansa J (2012) Real-time PCR to quantify composition of arbuscular mycorrhizal fungal communities–marker design, verification, calibration and field validation. Mol Ecol Resour 12:219–232. https://doi.org/10.1111/j.1755-0998.2011.03086.x
Větrovský T, Baldrian P, Morais D (2018) SEED 2: a user-friendly platform for amplicon high-throughput sequencing data analyses. Bioinformatics 34:2292–2294. https://doi.org/10.1093/bioinformatics/bty071
Worrich A, König S, Miltner A et al (2016) Mycelium-like networks increase bacterial dispersal, growth, and biodegradation in a model ecosystem at various water potentials. Appl Environ Microbiol 82:2902. https://doi.org/10.1128/aem.03901-15
Yang W, Li P, Rensing C et al (2019) Biomass, activity and structure of rhizosphere soil microbial community under different metallophytes in a mining site. Plant Soil 434:245–262. https://doi.org/10.1007/s11104-017-3546-9
Zhang M, Zeng H, Afzal MR et al (2022) BNI-release mechanisms in plant root systems: current status of understanding. Biol Fertil Soils 3:225–233. https://doi.org/10.1007/s00374-021-01568-y
Zhao X, Tian P, Sun Z et al (2022) Rhizosphere effects on soil organic carbon processes in terrestrial ecosystems: A meta-analysis. Geoderma 412:115739. https://doi.org/10.1016/j.geoderma.2022.115739
Acknowledgements
The research described here was funded by Czech Science Foundation (projects 21-07247S and 23-04690S) and further supported by the Czech Academy of Sciences (RVO 61388971).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Tatsuhiro Ezawa.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, D., Kotianová, M., Rozmoš, M. et al. Arbuscular mycorrhizal hyphae selectively suppress soil ammonia oxidizers – but probably not by production of biological nitrification inhibitors. Plant Soil 491, 627–643 (2023). https://doi.org/10.1007/s11104-023-06144-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-06144-x