Abstract
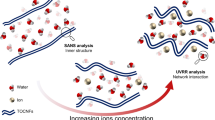
This work demonstrates for the first time the deconstruction of microfibrillated cellulose (MFC) into rod-like cellulose nanocrystals (CNCs) in concentrated low modulus sodium silicate solutions. To this aim, MFC suspensions at different concentrations were first treated in sodium hydroxide solutions and then amorphous silica powder was added. Optical microscopy and transmission electron microscopy observation showed how MFC was efficiently deconstructed into CNCs, evidencing the occurrence of a phase separation into an isotropic and mesogenic phase. The extracted CNCs were characterized by a remarkably higher length (600–1200 nm) in comparison with the plant-derived ones commonly reported in literature. FT-IR spectroscopy and 29Si MAS NMR confirmed that the Qn equilibrium of the suspended silicate species was affected, proportionally to the amount of MFC. It was also shown, that due to the excluded volume effect exerted by silicate anions, nematic or smectic ordering could be achieved for CNC concentrations far below the critical rod concentration predicted by the Doi-Edwards model.
Graphical abstract









Similar content being viewed by others
Abbreviations
- WG :
-
Water glass (aqueous sodium silicate solution)
- MFC :
-
Microfibrillated cellulose
- Na-MFC :
-
MFC treated in 10 M NaOH solution
- CNCs :
-
Cellulose nanocrystals
- n :
-
SiO2/Na2O molar ratio, modulus
- M sol :
-
Solid mass in the suspension obtained by weighting the solid residual (g)
- M WG :
-
WG mass obtained by stoichiometric calculations (g)
- M tot :
-
Suspension mass (g)
- M MFC :
-
Mass MFC (wet) (g)
- c s = 0.02:
-
Dry content of MFC
- w s :
-
Solid weight fraction = \(\frac{M_{sol} }{M_{tot} }\)
- w MFC :
-
MFC fibrils weight fraction = \(\frac{M_{MFC} \cdot c_{s} }{M_{WG} }\)
- w CNC :
-
The weight fraction of CNCs (wCNC) on the suspension mass (Mtot) = \(\frac{M_{MFC} \cdot c_{s} }{M_{tot} }\)
- ρ WG :
-
Density of a water glass suspension as a function of the solid content solution (g cm−3)
- ρ CNC :
-
Density of CNCs (g cm−3)
- MFC0 :
-
Pure water glass
- MFC1 :
-
WG/MFC suspension with wMFC = 0.01
- MFC2 :
-
WG/MFC suspension with wMFC = 0.02
- \(Q_z^n\) :
-
Silicate consisting of z interconnected Qn units
- w CNC = w MFC·w s :
-
CNCs weight fraction on the whole suspension mass
- ϕ CNC :
-
Volume fraction of CNCs
- ϕ WG :
-
Volume fraction of silicate anions
- L CNC :
-
CNC’s length (m)
- d CNC :
-
CNC’s diameter (m)
- ν = ϕ CNC/(π/4)·d 2 CNC L CNC :
-
Number of rods per unit volume of solution
- β :
-
Flow index
- K :
-
Consistency index
- \(\dot{\gamma }\) :
-
Shear rate (s−1)
- η 0 :
-
Zero shear viscosity (Pa s)
- η 0.01 :
-
Viscosity at shear rate = 0.01 s−1 (Pa s)
- η s :
-
Viscosity of the suspending medium (Pa s)
- σ y :
-
Yield stress (Pa)
- G′, G″:
-
Storage and loss moduli (Pa s)
- k B :
-
Boltzmann’s constant (J K−1)
- T :
-
Temperature (K)
- d* CNC :
-
Increased CNC’s diameter due to silicate adsorption (m)
- d rod :
-
Diameter of a generic rod (Dogic and Frenkel) (m)
- L rod :
-
Length of a generic rod (Dogic and Frenkel) (m)
- d WG :
-
Diameter of a spherical silicate anion (m)
- δ min :
-
Minimum interfibrillar distance (m)
- s :
-
± number of brushes/4
- D CNC :
-
Rotatory diffusivity of CNCs in diluted regime (m2 s−1)
- \(D_{CNC}^{\prime }\) :
-
Rotatory diffusivity of CNCs in semidiluted regime (m2 s−1)
- ω :
-
Frequency (Hz)
- ω c :
-
Crossover frequency (Hz)
References
Adams M, Dogic Z, Keller SL, Fraden S (1998) Entropically driven microphase transitions in of colloidal rods and spheres. Nature 393:349–352. https://doi.org/10.1038/30700
Angeli F, Villain O, Schuller S, Ispas S (2011) Insight into sodium silicate glass structural organization by multinuclear NMR combined with first-principles calculations. Geochim Cosmochim Acta 75:2453–2469. https://doi.org/10.1016/j.gca.2011.02.003
Araki J, Mishima S (2014) Steric stabilization of “charge-free” cellulose nanowhiskers by grafting of poly(ethylene glycol). Molecules 20:169–184. https://doi.org/10.3390/molecules20010169
Atalla R, VanderHart D (2011) Native cellulose: a composite of two distinct crystalline forms. Science (80-) 223:283–285. https://doi.org/10.1126/science.223.4633.283
Aveston J (1965) Hydrolysis of sodium silicate: ultracentrifugation in chloride solutions. J Chem Soc. https://doi.org/10.1039/jr9650004444
Axelrad P, Larson KM (2006) The structure and rheology of complex fluids. Eos Trans Am Geophys Union 87:227
Barnes HA (1995) A review of the slip (wall depletion) of polymer solutions, emulsions and particle suspensions in viscometers: its cause, character, and cure. J Non-Newton Fluid Mech 56:221–231. https://doi.org/10.1016/0377-0257(94)01282-M
Bechhoefer J, Lejcek L, Oswald P (1992) Facets of smectic A droplets I. Shape measurements. J Phys II 2:27. https://doi.org/10.1051/jp2:1992111>
Belton PS, Tanner SF, Cartier N, Chanzy H (1989) High-resolution solid-state carbon-13 nuclear magnetic resonance spectroscopy of tunicin, an animal cellulose. Macromolecules 22:1615–1617. https://doi.org/10.1021/ma00194a019
Bennington CPJ, Kerekes RJ, Grace JR (1990) The yield stress of fibre suspensions. Can J Chem Eng 68:748–757. https://doi.org/10.1002/cjce.5450680503
Bernal SA, Provis JL, Rose V, Mejía De Gutierrez R (2011) Evolution of binder structure in sodium silicate-activated slag-metakaolin blends. Cem Concr Compos 33:46–54. https://doi.org/10.1016/j.cemconcomp.2010.09.004
Bobrowsky A, Stypula B, Hutera A (2012) FTIR spectroscopy of water glass—the binder moulding modified by ZnO nanoparticles. Metalurgija 51:477–480
Brown AJ (1886) XLIII.—On an acetic ferment which forms cellulose. J Chem Soc Trans 49:432–439. https://doi.org/10.1039/CT8864900432
Budtova T, Navard P (2016) Cellulose in NaOH–water based solvents: a review. Cellulose 23:5–55. https://doi.org/10.1007/s10570-015-0779-8
Chen J, Cranton W, Fihn M (2012) Handbook of visual display technology. Springer, Berlin
Clayden NJ, Esposito S, Aronne A, Pernice P (1999) Solid state 27Al NMR and FTIR study of lanthanum aluminosilicate glasses. J Non Cryst Solids 258:11–19. https://doi.org/10.1016/S0022-3093(99)00555-4
Colby RH, Ober CK, Gillmor JR et al (1997) Smectic rheology. Rheol Acta 36:498–504. https://doi.org/10.1007/BF00368127
Dalby KN, King PL (2006) A new approach to determine and quantify structural units in silicate glasses using micro-reflectance Fourier-Transform infrared spectroscopy. Am Mineral 91:1783–1793. https://doi.org/10.2138/am.2006.2075
Demus D (1975) Schlieren textures in smectic liquid crystals. Krist und Tech 10:933–946. https://doi.org/10.1002/crat.19750100903
Dimas D, Giannopoulou I, Panias D (2009) Polymerization in sodium silicate solutions: a fundamental process in geopolymerization technology. J Mater Sci 44:3719–3730. https://doi.org/10.1007/s10853-009-3497-5
Ding S-Y, Himmel ME (2006) The maize primary cell wall microfibril: a new model derived from direct visualization. J Agric Food Chem 54:597–606. https://doi.org/10.1021/jf051851z
Dogic Z, Frenkel D, Fraden S (2000) Enhanced stability of layered phases in parallel hard spherocylinders due to addition of hard spheres. Phys Rev E 62:3925–3933. https://doi.org/10.1103/PhysRevE.62.3925
Elazzouzi-Hafraoui S, Nishiyama Y, Putaux J-L et al (2008) The shape and size distribution of crystalline nanoparticles prepared by acid hydrolysis of native cellulose. Biomacromol 9:57–65. https://doi.org/10.1021/bm700769p
Fengel D, Strobel C (1994) FTIR spectroscopic studies on the heterogeneous transformation of cellulose I into cellulose II. Acta Polym 45:319–324. https://doi.org/10.1002/actp.1994.010450406
Flauzino Neto WP, Putaux J-L, Mariano M et al (2016) Comprehensive morphological and structural investigation of cellulose I and II nanocrystals prepared by sulphuric acid hydrolysis. RSC Adv 6:76017–76027. https://doi.org/10.1039/C6RA16295A
Fujii S, Komura S, Lu CYD (2014) Structural rheology of the smectic phase. Materials (Basel) 7:5146–5168. https://doi.org/10.3390/ma7075146
Habibi Y, Lucia LA, Rojas OJ (2010) Cellulose nanocrystals: chemistry, self-assembly, and applications. Chem Rev 110:3479–3500. https://doi.org/10.1021/cr900339w
Halasz I, Agarwal M, Li R, Miller N (2010) What can vibrational spectroscopy tell about the structure of dissolved sodium silicates? Microporous Mesoporous Mater 135:74–81. https://doi.org/10.1016/J.MICROMESO.2010.06.013
Han J, Zhou C, Wu Y et al (2013) Self-assembling behavior of cellulose nanoparticles during freeze-drying: effect of suspension concentration, particle size, crystal structure, and surface charge. Biomacromol 14:1529–1540. https://doi.org/10.1021/bm4001734
Harris RK, Knight CT (1983) Silicon-29 nuclear magnetic resonance studies of aqueous silicate solutions. J Chem Soc Faraday Trans 2(79):1525–1538. https://doi.org/10.1002/mrc.1260241005
Heux L, Chauve G, Bonini C (2000) Nonflocculating and chiral-nematic self-ordering of cellulose microcrystals suspensions in nonpolar solvents. Langmuir 16:8210–8212. https://doi.org/10.1021/la9913957
Hoeger I, Rojas OJ, Efimenko K et al (2011) Ultrathin film coatings of aligned cellulose nanocrystals from a convective-shear assembly system and their surface mechanical properties. Soft Matter 7:1957. https://doi.org/10.1039/c0sm01113d
Hotta A, Terentjev EM (2003) Dynamic soft elasticity in monodomain nematic elastomers. Eur Phys J E 10:291–301. https://doi.org/10.1140/epje/i2002-10005-5
Jansson H, Bernin D, Ramser K (2015a) Silicate species of water glass and insights for alkali-activated green cement. AIP Adv 5:067103. https://doi.org/10.1063/1.4923371
Jansson H, Bernin D, Ramser K (2015b) Silicate species of water glass and insights for alkali-activated green cement. AIP Adv 5:067167. https://doi.org/10.1063/1.4923371
Kent JA, Bommaraju T, Barnicky SD (2017) Handbook of industrial chemistry and biotechnology. Springer International Publishing, Berlin
Klemm D, Kramer F, Moritz S et al (2011) Nanocelluloses: a new family of nature-based materials. Angew Chemie Int Ed 50:5438–5466. https://doi.org/10.1002/anie.201001273
Kolpak FJ, Blackwell J (1976) Determination of the structure of cellulose II. Macromolecules 9:273–278. https://doi.org/10.1021/ma60050a019
Kondo T (2005) Hydrogen bonds in cellulose and cellulose derivatives. In: Polysaccharides: structural diversity and functional versatility. https://doi.org/10.1201/9781420030822.ch3
Kondo T, Sawatari C (1996) A Fourier transform infra-red spectroscopic analysis of the character of hydrogen bonds in amorphous cellulose. Polymer (Guildf) 37:393–399. https://doi.org/10.1016/0032-3861(96)82908-9
Lago S, Cuetos A, De Olavide P, Rull LF (2004) Crowding effects and phase diagram of binary mixtures of rod-like and spherical particles. J Mol Recognit 17:417–425. https://doi.org/10.1002/jmr.704
Lavoine N, Desloges I, Dufresne A, Bras J (2012) Microfibrillated cellulose—its barrier properties and applications in cellulosic materials: a review. Carbohydr Polym 90:735–764. https://doi.org/10.1016/j.carbpol.2012.05.026
Mahler J, Sebald A (1995) Deconvolution of 29Si magic-angle spinning nuclear magnetic resonance spectra of silicate glasses revisited—some critical comments. Solid State Nucl Magn Reson 5:63–78. https://doi.org/10.1016/0926-2040(95)00027-N
McGarry SMA, Hazel JF (1965) Electron microscopy of labile alkali silicate solutions. J Colloid Sci 20:72–80
Miao C, Hamad WY (2013) Cellulose reinforced polymer composites and nanocomposites: a critical review. Cellulose 20:2221–2262. https://doi.org/10.1007/s10570-013-0007-3
Moberg T, Rigdahl M (2012) On the viscoelastic properties of microfibrillated cellulose (MFC) suspensions. Annu Trans Nord Rheol Soc 20:123–130
Morais JPS, Rosa M de F, Filho M de sá M, Nascimento LD (2013) Extraction and characterization of nanocellulose structures from raw cotton linter. Carbohydr Polym 91:229–235. https://doi.org/10.1016/j.carbpol.2012.08.010
Moreira L, Fulchiron R, Seytre G et al (2010) Aggregation of carbon nanotubes in semidilute suspension. Macromolecules 43:1467–1472. https://doi.org/10.1021/ma902433v
Nehring J, Saupe A (1972) On the schlieren texture in nematic and smectic liquid crystals. J Chem Soc Faraday Trans 2(68):1. https://doi.org/10.1039/f29726800001
Nordström J, Nilsson E, Jarvol P et al (2011) Concentration- and pH-dependence of highly alkaline sodium silicate solutions. J Colloid Interface Sci 356:37–45. https://doi.org/10.1016/j.jcis.2010.12.085
Oh YS, Il Yoo D, Shin Y et al (2005) Crystalline structure analysis of cellulose treated with sodium hydroxide and carbon dioxide by means of X-ray diffraction and FTIR spectroscopy. Carbohydr Res 340:2376–2391. https://doi.org/10.1016/j.carres.2005.08.007
Pääkkö M, Ankerfors M, Kosonen H et al (2007) Enzymatic hydrolysis combined with mechanical shearing and high-pressure homogenization for nanoscale cellulose fibrils and strong gels. Biomacromol 8:1934–1941. https://doi.org/10.1021/bm061215p
Pullawan T, Wilkinson AN, Eichhorn SJ (2012) Influence of magnetic field alignment of cellulose whiskers on the mechanics of all-cellulose nanocomposites. Biomacromol 13:2528–2536. https://doi.org/10.1021/bm300746r
Rahmat N, Hamzah F, Sahiron N et al (2016) Sodium silicate as source of silica for synthesis of mesoporous SBA-15. IOP Conf Ser Mater Sci Eng 133:1–9. https://doi.org/10.1088/1757-899X/133/1/012011
Revol JF, Bradford H, Giasson J et al (1992) Helicoidal self-ordering of cellulose microfibrils in aqueous suspension. Int J Biol Macromol 14:170–172. https://doi.org/10.1016/S0141-8130(05)80008-X
Sarawade PB, Kim J-K, Hilonga A, Kim HT (2010) Production of low-density sodium silicate-based hydrophobic silica aerogel beads by a novel fast gelation process and ambient pressure drying process. Solid State Sci 12:911–918. https://doi.org/10.1016/j.solidstatesciences.2010.01.032
Sierra L, Lopez B, Guth J-L (2000) Preparation of mesoporous silica particles with controlled morphology from sodium silicate solutions and a non-ionic surfactant at pH values between 2 and 6. Microporous Mesoporous Mater 39:519–527. https://doi.org/10.1016/S1387-1811(00)00227-4
Siqueira G, Bras J, Dufresne A (2010) New process of chemical grafting of cellulose nanoparticles with a long chain isocyanate. Langmuir 26:402–411. https://doi.org/10.1021/la9028595
Sugiyama J, Persson J, Chanzy H (1991) Combined infrared and electron diffraction study of the polymorphism of native celluloses. Macromolecules 24:2461–2466. https://doi.org/10.1021/ma00009a050
Sugiyama J, Chanzy H, Maret G (1992) Orientation of cellulose microcrystals by strong magnetic fields. Macromolecules 25:4232–4234. https://doi.org/10.1021/ma00042a032
Svensson IL, Sjöberg S, Öhman L-O (1986) Polysilicate equilibria in concentrated sodium silicate solutions. J Chem Soc Faraday Trans 1 Phys Chem Condens Phases 82:3635–3646. https://doi.org/10.1039/f19868203635
Taylor WR (1990) Application of infrared spectroscopy to studies of silicate glass structure: examples from the melilite glasses and the systems Na2O-SiO2 and Na2O-Al2O3-SiO2. J Earth Syst Sci 99:99–117. https://doi.org/10.1007/BF02871899
Vidal L, Joussein E, Colas M et al (2016) Controlling the reactivity of silicate solutions: a FTIR, Raman and NMR study. Colloids Surfaces A Physicochem Eng Asp 503:101–109. https://doi.org/10.1016/j.colsurfa.2016.05.039
Wang Y (2008) Cellulose fiber dissolution in sodium hydroxide solution at low temperature: dissolution kinetics and solubility improvement. Georgia Institute of Technology, Atlanta
Wang S, Wang DK, Smart S, Diniz Da Costa JC (2015) Ternary phase-separation investigation of sol-gel derived silica from ethyl silicate 40. Sci Rep 5:1–11. https://doi.org/10.1038/srep14560
Yang X, Zhu W, Yang Q (2008) The viscosity properties of sodium silicate solutions. J Solut Chem 37:73–83. https://doi.org/10.1007/s10953-007-9214-6
Yue Y, Han G, Wu Q (2013) Transitional properties of cotton fibers from cellulose I to cellulose II structure. BioResources 8:6460–6471. https://doi.org/10.15376/biores.8.4.6460-6471
Zhang P, Dunlap C, Florian P et al (1996) Silicon site distributions in an alkali silicate glass derived by two-dimensional 29Si nuclear magnetic resonance. J Non Cryst Solids 204:294–300. https://doi.org/10.1016/S0022-3093(96)00601-1
Acknowledgments
This work was realised at CEITEC IPM—Central European Institute of Technology with research infrastructure supported by the project CZ.1.05/1.1.00/02.0068 financed from European Regional Development Fund. The support from the project Materials Research Centre at FCH BUT- Sustainability and Development REG LO1211 and the project LO1219 with financial support from the National Program for Sustainability I (Ministry of Education, Youth and Sports) is gratefully acknowledged. CIISB research infrastructure project LM2015043 funded by MEYS CR at the CF Biomolecular Interactions and Crystallization is also gratefully acknowledged. Finally, a special mention goes to Dr. Jiří Smilek, Dr. Alberto Viani, Dr. Jiri Holas and Prof. Giulio Malucelli for the support provided for the rheological tests, DIC microscopy, XRD analysis and data interpretation, respectively.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bertolla, L., Dlouhý, I., Bartoničková, E. et al. Deconstruction of microfibrillated cellulose into nanocrystalline cellulose rods and mesogenic phase formation in concentrated low-modulus sodium silicate solutions. Cellulose 26, 4325–4344 (2019). https://doi.org/10.1007/s10570-019-02364-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-019-02364-6