Abstract
This study compares two types of bioresorptive cellulose, i.e., calcium-sodium salt of oxidized cellulose (OC) and sodium salt of carboxymethylcellulose (CMC). It investigates which type would be preferable as an implant material in terms of biocompatibility, biomechanical and biological properties, and also in terms of its behavior in combination with collagen fibrils (Col) in composite Col/OC or Col/CMC scaffolds. OC significantly supported the stiffness and elasticity of Col fibrils, whereas CMC significantly reduced these properties. OC also enabled a strong interaction with Col fibrils even in a moist environment, accompanied by a significant drop in elastic modulus. The addition of cellulose did not significantly influence scaffold porosity; however, changes in surface morphology and the lower swelling capacity of OC, with a degree of oxidation of its chains between 16 and 24%, supported the idea of improved cell-material interaction. The elasticity and the stiffness of Col/OC guided human adipose-derived stem cells (hADSCs) to significantly higher adhesion, proliferation, and metabolic activity. On the contrary, the Col/CMC provided only limited mechanical support for the cells and inhibited their attachment and proliferation, although without any signs of cytotoxicity. This phenomenon could be used for future control of the differentiation of hADSCs towards a desired phenotype to generate advanced tissue replacements using modern methods of tissue engineering. The oxidation of cellulose resulted in a firmer scaffolding material, as required in vascular or skin tissue engineering. CMC is suitable for moist wound healing, e.g. as a mucoadhesive gel, where cell adhesion is not desirable.
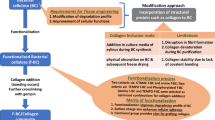
Graphical abstract













Similar content being viewed by others
Data availability
Without restrictions.
References
Abdollahi M, Damirchi S, Shafafi M, Rezaei M, Ariaii P (2019) Carboxymethyl cellulose-agar biocomposite film activated with summer savory essential oil as an antimicrobial agent. Int J Biol Macromol 126:561–568. https://doi.org/10.1016/j.ijbiomac.2018.12.115
Abitbol T, Kam D, Levi-Kalisman Y, Gray DG, Shoseyov O (2018) Surface charge influence on the phase separation and viscosity of cellulose nanocrystals. Langmuir 34(13):3925–3933. https://doi.org/10.1021/acs.langmuir.7b04127
Akhtar R, Sherratt MJ, Cruickshank JK, Derby B (2011) Characterizing the elastic properties of tissues. Mater Today. https://doi.org/10.1016/S1369-7021(11)70059-1
Alavi M, Nokhodchi A (2020) Antimicrobial and wound treatment aspects of micro- and nanoformulations of carboxymethyl, dialdehyde, and TEMPO-oxidized derivatives of cellulose: recent advances. Macromol Biosci 20(4):1900362. https://doi.org/10.1002/mabi.201900362
An Y, Cui B, Wang Y, Jin W, Geng X, Yan X, Li B (2014) Functional properties of ovalbumin glycosylated with carboxymethyl cellulose of different substitution degree. Food Hydrocoll 40:1–8. https://doi.org/10.1016/j.foodhyd.2014.01.028
Babrnáková J, Pavliňáková V, Brtníková J, Sedláček P, Prosecká E, Rampichová M, Filová E, Hearnden V, Vojtová L (2019) Synergistic effect of bovine platelet lysate and various polysaccharides on the biological properties of collagen-based scaffolds for tissue engineering: Scaffold preparation, chemo-physical characterization, in vitro and ex ovo evaluation. Mater Sci Eng C. https://doi.org/10.1016/j.msec.2019.02.092
Bacakova L, Zarubova J, Travnickova M, Musilkova J, Pajorova J, Slepicka P, Kasalkova NS, Svorcik V, Kolska Z, Motarjemi H, Molitor M (2018) Stem cells: their source, potency and use in regenerative therapies with focus on adipose-derived stem cells—a review. Biotechnol Adv. https://doi.org/10.1016/j.biotechadv.2018.03.011
Bäckdahl H, Helenius G, Bodin A, Nannmark U, Johansson BR, Risberg B, Gatenholm P (2006) Mechanical properties of bacterial cellulose and interactions with smooth muscle cells. Biomaterials 27(9):2141–2149. https://doi.org/10.1016/j.biomaterials.2005.10.026
Basta AH, Lotfy VF, Mahmoud K, Abdelwahed NAM (2020) Synthesis and evaluation of protein-based biopolymer in production of silver nanoparticles as bioactive compound versus carbohydrates-based biopolymers. R Soc Open Sci 7(10):200928. https://doi.org/10.1098/rsos.200928
Basu P, Narendrakumar U, Arunachalam R, Devi S, Manjubala I (2018) Characterization and evaluation of carboxymethyl cellulose-based films for healing of full-thickness wounds in normal and diabetic rats. ACS Omega 3(10):12622–12632. https://doi.org/10.1021/acsomega.8b02015
Bat E, Kothman BHM, Higuera GA, van Blitterswijk CA, Feijen J, Grijpma DW (2010) Ultraviolet light crosslinking of poly(trimethylene carbonate) for elastomeric tissue engineering scaffolds. Biomaterials 31(33):8696–8705. https://doi.org/10.1016/j.biomaterials.2010.07.102
Chang Ch, He M, Zhou J, Zhang L (2011) Swelling behaviors of pH- and salt-responsive cellulose-based hydrogels. Macromolecules 44(6):1642–1648. https://doi.org/10.1021/ma102801f
Chen H, Fan M (2007) Chitosan/carboxymethyl cellulose polyelectrolyte complex scaffolds for pulp cells regeneration. J Bioact Compat Polym 22(5):475–491. https://doi.org/10.1177/0883911507081329
Clarke SA, Hoskins NL, Jordan GR, Henderson SA, Marsh DR (2007) In vitro testing of advanced JAX™ bone void filler system: species differences in the response of bone marrow stromal cells to β tri-calcium phosphate and carboxymethylcellulose gel. J Mater Sci: Mater Med 18(12):2283–2290. https://doi.org/10.1007/s10856-007-3099-1
Coenen AMJ, Bernaerts KV, Harings JAW, Jockenhoevel S, Ghazanfari S (2018) Elastic materials for tissue engineering applications: natural, synthetic, and hybrid polymers. Acta Biomater 79:60–82. https://doi.org/10.1016/j.actbio.2018.08.027
Copes F, Pien N, van Vlierberghe S, Boccafoschi F, Mantovani D (2019) Collagen-based tissue engineering strategies for vascular medicine. Front Bioeng Biotechnol. https://doi.org/10.3389/fbioe.2019.00166
Courtenay JC, Johns MA, Galembeck F, Deneke C, Lanzoni EM, Costa CA, Scott JL, Sharma RI (2017) Surface modified cellulose scaffolds for tissue engineering. Cellulose 24(1):253–267. https://doi.org/10.1007/s10570-016-1111-y
Courtenay JC, Deneke C, Lanzoni EM, Costa CA, Bae Y, Scott JL, Sharma RI (2018) Modulating cell response on cellulose surfaces; tunable attachment and scaffold mechanics. Cellulose 25(2):925–940. https://doi.org/10.1007/s10570-017-1612-3
Crichton ML, Donose BC, Chen X, Raphael AP, Huang H, Kendall MAF (2011) The viscoelastic, hyperelastic and scale dependent behaviour of freshly excised individual skin layers. Biomaterials. https://doi.org/10.1016/j.biomaterials.2011.03.012
Dadsetan M, Pumberger M, Casper ME, Shogren K, Giuliani M, Ruesink T, Hefferan TE, Currier BL, Yaszemski MJ (2011) The effects of fixed electrical charge on chondrocyte behavior. Acta Biomater 7(5):2080–2090. https://doi.org/10.1016/j.actbio.2011.01.012
de Lima GG, de Lima DWF, de Oliveira MJA, Lugão AB, Alcântara MTS, Devine DM, de Sá MJC (2018) Synthesis and in vivo behavior of PVP/CMC/agar hydrogel membranes impregnated with silver nanoparticles for wound healing applications. ACS Appl Bio Mater 1(6):1842–1852. https://doi.org/10.1021/acsabm.8b00369
Diaz-Gomez L, Gonzalez-Prada I, Millan R, da Silva-Candal A, Bugallo-Casal A, Campos F, Concheiro A, Alvarez-Lorenzo C (2022) 3D printed carboxymethyl cellulose scaffolds for autologous growth factors delivery in wound healing. Carbohydr Polym 278:118924. https://doi.org/10.1016/j.carbpol.2021.118924
Dong C, Lv Y (2016) Application of collagen scaffold in tissue engineering: recent advances and new perspectives. Polymers. https://doi.org/10.3390/polym8020042
Dorazilová J, Muchová J, Šmerková K, Kočiová S, Diviš P, Kopel P, Veselý R, Pavliňáková V, Adam V, Vojtová L (2020) Synergistic effect of chitosan and selenium nanoparticles on biodegradation and antibacterial properties of collagenous scaffolds designed for infected burn wounds. Nanomaterials 10(10):1971. https://doi.org/10.3390/nano10101971
Dutta SD, Patel DK, Lim KT (2019) Functional cellulose-based hydrogels as extracellular matrices for tissue engineering. J Biol Eng. https://doi.org/10.1186/s13036-019-0177-0
Engler A, Bacakova L, Newman C, Hategan A, Griffin M, Discher D (2004) Substrate compliance versus ligand density in cell on gel responses. Biophys J. https://doi.org/10.1016/S0006-3495(04)74140-5
Fernández J, Etxeberria A, Sarasua JR (2012) Synthesis, structure and properties of poly(L-lactide-co-ε-caprolactone) statistical copolymers. J Mech Behav Biomed Mater. https://doi.org/10.1016/j.jmbbm.2012.01.003
Feng X, Ullah N, Wang X, Sun X, Li C, Bai Y, Chen L, Li Z (2015) Characterization of bacterial cellulose by gluconacetobacter CGMCC 3917. J Food Sci 80(10):E2217–E2227. https://doi.org/10.1111/1750-3841.13010
Fink H, Ahrenstedt L, Bodin A, Brumer H, Gatenholm P, Krettek A, Risberg B (2011) Bacterial cellulose modified with xyloglucan bearing the adhesion peptide RGD promotes endothelial cell adhesion and metabolism-a promising modification for vascular grafts. J Tissue Eng Regen Med 5(6):454–463. https://doi.org/10.1002/term.334
Ghasemi-Mobarakeh L, Prabhakaran MP, Tian L, Shamirzaei-Jeshvaghani E, Dehghani L, Ramakrishna S (2015) Structural properties of scaffolds: crucial parameters towards stem cells differentiation. World J Stem Cells 7(4):728–744. https://doi.org/10.4252/wjsc.v7.i4.728
Grant CA, Brockwell DJ, Radford SE, Thomson NH (2008) Effects of hydration on the mechanical response of individual collagen fibrils. Appl Phys Lett 10(1063/1):2937001
Griffin M, Premakumar Y, Seifalian A, Butler PE, Szarko M (2016) Biomechanical characterization of human soft tissues using indentation and tensile testing. J vis Exp. https://doi.org/10.3791/54872
Guo B, Ma PX (2014) Synthetic biodegradable functional polymers for tissue engineering: a brief review. Sci China Chem. https://doi.org/10.1007/s11426-014-5086-y
Heinze T, Pfeiffer K (1999) Studies on the synthesis and characterization of carboxymethylcellulose. Macromol Mater Eng 266(1):37–45. https://doi.org/10.1002/(SICI)1522-9505(19990501)266:1%3c37::AID-APMC37%3e3.0.CO;2-Z
Hollabaugh CB, Burt LH, Walsh AP (1945) Carboxymethylcellulose. Uses and applications. Ind Eng Chem. https://doi.org/10.1021/ie50430a015
Jantrawut P, Bunrueangtha J, Suerthong J, Kantrong N (2019) Fabrication and characterization of low methoxyl pectin/gelatin/carboxymethyl cellulose absorbent hydrogel film for wound dressing applications. Materials 12(10):1628. https://doi.org/10.3390/ma12101628
Joy J, Pereira J, Aid-Launais R, Pavon-Djavid G, Ray AR, Letourneur D, Meddahi-Pellé A, Gupta B (2018) Gelatin—oxidized carboxymethyl cellulose blend based tubular electrospun scaffold for vascular tissue engineering. Int J Biol Macromol 107:1922–1935. https://doi.org/10.1016/j.ijbiomac.2017.10.071
Katoh K, Tanabe T, Yamauchi K (2004) Novel approach to fabricate keratin sponge scaffolds with controlled pore size and porosity. Biomaterials 25(18):4255–4262. https://doi.org/10.1016/j.biomaterials.2003.11.018
Ke Y, Liu GS, Wang JH, Xue W, Du C, Wu G (2014) Preparation of carboxymethyl cellulose based microgels for cell encapsulation. Express Polym Lett. https://doi.org/10.3144/expresspolymlett.2014.85
Ke Y, Liu C, Wang Y, Xiao M, Fan J, Fu P, Wang S, Wu G (2018) Cell-loaded carboxymethylcellulose microspheres sustain viability and proliferation of ATDC5 cells. Artif Cells Nanomed Biotechnol 46(sup2):140–151. https://doi.org/10.1080/21691401.2018.1452751
Kilic Bektas C, Kimiz I, Sendemir A, Hasirci V, Hasirci N (2018) A bilayer scaffold prepared from collagen and carboxymethyl cellulose for skin tissue engineering applications. J Biomater Sci Polym Ed 29(14):1764–1784. https://doi.org/10.1080/09205063.2018.1498718
Kwon IK, Park KD, Choi SW, Lee SH, Lee EB, Na JS, Kim SH, Kim YH (2001) Fibroblast culture on surface-modified poly (glycolide-co-ε-caprolactone) scaffold for soft tissue regeneration. J Biomater Sci Polym Ed 12(10):1147–1160. https://doi.org/10.1163/15685620152691904
Lepcio P, Svatík J, Režnáková E, Zicha D, Lesser AJ, Ondreáš F (2022) Anisotropic solid-state PLA foaming templated by crystal phase pre-oriented with 3D printing: cell supporting structures with directional capillary transfer function. J Mater Chem B 10(15):2889–2898. https://doi.org/10.1039/D1TB02133H
Levanič J, Šenk VP, Nadrah P, Poljanšek I, Oven P, Haapala A (2020) Analyzing TEMPO-oxidized cellulose fiber morphology: new insights into optimization of the oxidation process and nanocellulose dispersion quality. ACS Sustain Chem Eng 8(48):17752–17762. https://doi.org/10.1021/acssuschemeng.0c05989
Li L (2021) Nanoscale chargé density measurement in liquid with AFM. Dissertation, Case Western Reserve University
Li N, Zhong Q (2021) Effects of polysaccharide charge density on the structure and stability of carboxymethylcellulose-casein nanocomplexes at pH 4.5 prepared with and without a pH-cycle. Food Hydrocoll 117:106718. https://doi.org/10.1016/j.foodhyd.2021.106718
Li M, Li F, Wang T, Zhao L, Shi Y (2020) Fabrication of carboxymethylcellulose hydrogel containing β-cyclodextrin–eugenol inclusion complexes for promoting diabetic wound healing. J Biomater Appl 34(6):851–863. https://doi.org/10.1177/0885328219873254
Lin N, Bruzzese C, Dufresne A (2012) TEMPO-oxidized nanocellulose participating as crosslinking aid for alginate-based sponges. ACS Appl Mater Interfaces 4(9):4948–4959. https://doi.org/10.1021/am301325r
Liu J, Zheng H, Poh PSP, Machens HG, Schilling AF (2015) Hydrogels for engineering of perfusable vascular networks. Int J Mol Sci. https://doi.org/10.3390/ijms160715997
Malm CJ, Risberg B, Bodin A, Bäckdahl H, Johansson BR, Gatenholm P, Jeppsson A (2012) Small calibre biosynthetic bacterial cellulose blood vessels: 13-months patency in a sheep model. Scand Cardiovasc J Suppl 46(1):57–62. https://doi.org/10.3109/14017431.2011.623788
Martina B, Kateřina K, Miloslava R, Jan G, Ruta M (2009) Oxycellulose: significant characteristics in relation to its pharmaceutical and medical applications. Adv Polym Technol. https://doi.org/10.1002/adv.20161
Mirtaghavi A, Luo J, Muthuraj R (2020) Recent advances in porous 3D cellulose aerogels for tissue engineering applications: a review. J Compos Sci 4(4):152. https://doi.org/10.3390/jcs4040152
Moradian M, Alam MN, van de Ven TGM (2021) Influence of carboxyl charge density on properties of extruded cellulose films. Ind Eng Chem Res 60(38):13756–13763. https://doi.org/10.1021/acs.iecr.1c01716
Nishi Y, Uryu M, Yamanaka S, Watanabe K, Kitamura N, Iguchi M, Mitsuhashi S (1990) The structure and mechanical properties of sheets prepared from bacterial cellulose—part 2 Improvement of the mechanical properties of sheets and their applicability to diaphragms of electroacoustic transducers. J Mater Sci. https://doi.org/10.1007/BF00584917
Novotna K, Havelka P, Sopuch T, Kolarova K, Vosmanska V, Lisa V, Svorcik V, Bacakova L (2013) Cellulose-based materials as scaffolds for tissue engineering. Cellulose. https://doi.org/10.1007/s10570-013-0006-4
Oliveira RN, Moreira APD, da Silva Moreira Thiré RM, Quilty B, Passos TM, Simon P, Mancini MC, McGuinness GB (2017) Absorbent polyvinyl alcohol-sodium carboxymethyl cellulose hydrogels for propolis delivery in wound healing applications. Polym Eng Sci. https://doi.org/10.1002/pen.24500
Pajorova J, Skogberg A, Hadraba D, Broz A, Travnickova M, Zikmundova M, Honkanen M, Hannula M, Lahtinen P, Tomkova M, Bacakova L, Kallio P (2020) Cellulose mesh with charged nanocellulose coatings as a promising carrier of skin and stem cells for regenerative applications. Biomacromol. https://doi.org/10.1021/acs.biomac.0c01097
Paprskářová A, Suchý P, Chalupová M, Michlovská L, Klusáková J, Sopuch T, Vojtová L (2021) Evaluation and comparison of structurally different cellulose-based hemostatic agents in a rat kidney model. Cellulose. https://doi.org/10.1007/s10570-021-04104-1
Parenteau-Bareil R, Gauvin R, Berthod F (2010) Collagen-based biomaterials for tissue engineering applications. Materials. https://doi.org/10.3390/ma3031863
Park JS, Cha SJ, Kim BG, Choi YS, Kwon GY, Kang H, An SS (2011) An assessment of the effects of a hyaluronan-based solution on reduction of postsurgical adhesion formation in rats: a comparative study of hyaluronan-based solution and two film barriers. J Surg Res 168(1):49–55. https://doi.org/10.1016/j.jss.2009.09.025
Pêgo AP, Grijpma DW, Feijen J (2003) Enhanced mechanical properties of 1,3-trimethylene carbonate polymers and networks. Polymer 44(21):6495–6504. https://doi.org/10.1016/S0032-3861(03)00668-2
Qiu H, Zhu Y, Sun Z, Trzeciakowski JP, Gansner M, Depre C, Resuello RRG, Natividad FF, Hunter WC, Genin GM, Elson EL, Vatner DE, Meininger GA, Vatner SF (2010) Short communication: vascular smooth muscle cell stiffness as a mechanism for increased aortic stiffness with aging. Circ Res. https://doi.org/10.1161/CIRCRESAHA.110.221846
Ranjbar-Mohammadi M, Arab-Bafrani Z, Karimi F, Javid N (2021) Designing hybrid nanofibers based on keratin-poly (vinyl alcohol) and poly (Ɛ-caprolactone) for application as wound dressing. J Ind Text. https://doi.org/10.1177/1528083721988978
Rees A, Powell LC, Chinga-Carrasco G, Gethin DT, Syverud K, Hill KE, Thomas DW (2015) 3D bioprinting of carboxymethylated-periodate oxidized nanocellulose constructs for wound dressing applications. Biomed Res Int 2015:1–7. https://doi.org/10.1155/2015/925757
Ruszczak Z (2003) Effect of collagen matrices on dermal wound healing. Adv Drug Deliv Rev. https://doi.org/10.1016/j.addr.2003.08.003
Shoulders MD, Raines RT (2009) Collagen structure and stability. Annu Rev Biochem. https://doi.org/10.1146/annurev.biochem.77.032207.120833
Silvestre GH, Pinto LO, Bernardes JS, Miwa RH, Fazzio A (2021) Disassembly of TEMPO-oxidized cellulose fibers: intersheet and interchain interactions in the isolation of nanofibers and unitary chains. J Phys Chem B 125(14):3717–3724. https://doi.org/10.1021/acs.jpcb.1c01928
Sloviková A, Vojtová L, Jančař J (2008) Preparation and modification of collagen-based porous scaffold for tissue engineering. Chem Pap. https://doi.org/10.2478/s11696-008-0045-8
Snowhill PB, Silver FH (2005) A mechanical model of porcine vascular tissues-part II: Stress-strain and mechanical properties of juvenile porcine blood vessels. Cardiovasc Eng. https://doi.org/10.1007/s10558-005-9070-1
Song Y, Kamphuis MMJ, Zhang Z, Sterk LMTh, Vermes I, Poot AA, Feijen J, Grijpma DW (2010) Flexible and elastic porous poly(trimethylene carbonate) structures for use in vascular tissue engineering. Acta Biomater 6(4):1269–1277. https://doi.org/10.1016/j.actbio.2009.10.002
Spangler D, Rothenburger S, Nguyen K, Jampani H, Weiss S, Bhende S (2003) In vitro antimicrobial activity of oxidized regenerated cellulose against antibiotic-resistant microorganisms. Surg Infect 4(3):255–262. https://doi.org/10.1089/109629603322419599
Steele TWJ, Huang ChL, Nguyen E, Sarig U, Kumar S, Widjaja E, Loo JSC et al (2013) Collagen–cellulose composite thin films that mimic soft-tissue and allow stem-cell orientation. J Mater Sci 24(8):2013–2027. https://doi.org/10.1007/s10856-013-4940-3
Stojic M, López V, Montero A, Quílez C, de Aranda IG, Vojtova L, Luis Jorcano J, Velasco D (2019) Skin tissue engineering. Biom Skin Rep Regen. https://doi.org/10.1016/B978-0-08-102546-8.00003-0
Švachová V, Vojtová L, Pavliňák D, Vojtek L, Sedláková V, Hyršl P, Alberti M, Jaroš J, Hampl A, Jančář J (2016) Novel electrospun gelatin/oxycellulose nanofibers as a suitable platform for lung disease modeling. Mater Sci Eng C. https://doi.org/10.1016/j.msec.2016.05.059
Svatík J, Lepcio P, Ondreáš F, Zárybnická K, Zbončák M, Menčík P, Jančář J (2021) PLA toughening via bamboo-inspired 3D printed structural design. Polym Test 104. https://doi.org/10.1016/j.polymertesting.2021.107405
Tang Z, Li W, Lin X, Xiao H, Miao Q, Huang L, Chen L, Wu H (2017) TEMPO-oxidized cellulose with high degree of oxidation. Polymers 9(9):421. https://doi.org/10.3390/polym9090421
Teti G, Salvatore V, Focaroli S, Durante S, Mazzotti A, Dicarlo M, Mattioli-Belmonte M, Orsini G (2015) In vitro osteogenic and odontogenic differentiation of human dental pulp stem cells seeded on carboxymethyl cellulose-hydroxyapatite hybrid hydrogel. Front Physiol. https://doi.org/10.3389/fphys.2015.00297
Thein-Han WW, Saikhun J, Pholpramoo C, Misra RDK, Kitiyanant Y (2009) Chitosan-gelatin scaffolds for tissue engineering: physico-chemical properties and biological response of buffalo embryonic stem cells and transfectant of GFP-buffalo embryonic stem cells. Acta Biomater. https://doi.org/10.1016/j.actbio.2009.05.012
Travnicková M, Pajorova J, Zarubova J, Krocilova N, Molitor M, Bacakova L (2020) The influence of negative pressure and of the harvesting site on the characteristics of human adipose tissue-derived stromal cells from lipoaspirates. Stem Cells Int. https://doi.org/10.1155/2020/1016231
Wagenhäuser MU, Mulorz J, Ibing W, Simon F, Spin JM, Schelzig H, Oberhuber A (2016) Oxidized (non)-regenerated cellulose affects fundamental cellular processes of wound healing. Sci Rep 6(1):32238. https://doi.org/10.1038/srep32238
Wei J, Wang B, Li Z, Wu Z, Zhang M, Sheng N, Liang Q, Wang H, Chen S (2020) A 3D-printable TEMPO-oxidized bacterial cellulose/alginate hydrogel with enhanced stability via nanoclay incorporation. Carbohydr Polym 238:116207. https://doi.org/10.1016/j.carbpol.2020.116207
Wiegand C, Abel M, Hipler UC, Elsner P (2019) Effect of non-adhering dressings on promotion of fibroblast proliferation and wound healing in vitro. Sci Rep 9(1):4320. https://doi.org/10.1038/s41598-019-40921-y
Wippermann J, Schumann D, Klemm D, Kosmehl H, Salehi-Gelani S, Wahlers T (2009) Preliminary results of small arterial substitute performed with a new cylindrical biomaterial composed of bacterial cellulose. Eur J Vasc Endovasc Surg 37(5):592–596. https://doi.org/10.1016/j.ejvs.2009.01.007
Wiwatwongwana F, Promma N (2019) Characterization of gelatin-carboxymethylcellulose scaffolds. Int J Mater Mech Manuf 7(1):12–15. https://doi.org/10.18178/ijmmm.2019.7.1.420
Wong TW, Ramli NA (2014) Carboxymethylcellulose film for bacterial wound infection control and healing. Carbohydr Polym 112:367–375. https://doi.org/10.1016/j.carbpol.2014.06.002
Wu Q, Li X, Fu S, Li Q, Wang S (2017) Estimation of aspect ratio of cellulose nanocrystals by viscosity measurement: influence of surface charge density and NaCl concentration. Cellulose 24(8):3255–3264. https://doi.org/10.1007/s10570-017-1341-7
Wu Y, Zhang X, Qiu D, Pei Y, Li Y, Li B, Liu S (2021) Effect of surface charge density of bacterial cellulose nanofibrils on the rheology property of O/W Pickering emulsions. Food Hydrocoll 120:106944. https://doi.org/10.1016/j.foodhyd.2021.106944
Xiong W, Ren C, Tian M, Yang X, Li J, Li B (2017) Complex coacervation of ovalbumin-carboxymethylcellulose assessed by isothermal titration calorimeter and rheology: Effect of ionic strength and charge density of polysaccharide. Food Hydrocoll 73:41–50. https://doi.org/10.1016/j.foodhyd.2017.06.031
Zhu J, Marchant RE (2011) Design properties of hydrogel tissue-engineering scaffolds. Expert Rev Med Devices. https://doi.org/10.1586/erd.11.27
Zimnitsky DS, Yurkshtovich TL, Bychkovsky PM (2004) Synthesis and characterization of oxidized cellulose. J Polym Sci A 42(19):4785–4791. https://doi.org/10.1002/pola.20302
Zimnitsky DS, Yurkshtovich TL, Bychkovsky PM (2005) Multilayer adsorption of amino acids on oxidized cellulose. J Colloid Interface Sci 285(2):502–508. https://doi.org/10.1016/j.jcis.2004.12.021
Acknowledgments
We thank Petr Lepcio for useful consultations on charge density and for correcting the biomechanical aspects of this paper, and Zuzana Kadlecová for measurement of Zeta potential. CzechNanoLab project LM2018110 funded by MEYS CR is gratefully acknowledged for the financial support for measurements/sample fabrication at CEITEC Nano Research Infrastructure. Mr. Robin Healey (Czech Technical University, Prague) is gratefully acknowledged for his language revision of the manuscript.
Funding
The material aspects of this work were supported by the Ministry of Health of the Czech Republic under project no. NV 19-0500214, and the biological aspects were supported by the Czech Science Foundation (Grant No. 20-01641S). All rights reserved. We acknowledge the Light Microscopy Core Facility, IMG CAS, Prague, Czech Republic, supported by MEYS (LM2018129, CZ.02.1.01/0.0/0.0/18_046/0016045) and RVO: 68378050-KAV-NPUI, for their support with the confocal image analysis presented here.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by KK, MT, JP and MK. SEM pictures were provided by KH. Material preparation for biological analysis is attributed to JD. Confocal microscopy photography and analysis provided MT. Methodology and supervision is attributed to LV from the material part and LB from the biological part. The first draft of the manuscript was written by KK. Authors who commented on previous versions of the manuscript and did additional changes: MT, JD. Who critically reviewed the manuscript LB and LV. PP reviewed its mechanical part. Funding acquisition: LV, LB. All authors read and approved the final manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kacvinská, K., Trávníčková, M., Vojtová, L. et al. Porous cellulose-collagen scaffolds for soft tissue regeneration: influence of cellulose derivatives on mechanical properties and compatibility with adipose-derived stem cells. Cellulose 29, 8329–8351 (2022). https://doi.org/10.1007/s10570-022-04759-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-022-04759-4