Abstract
Main conclusion
Chenopodium ficifolium flowered under long days despite much lower expression of FLOWERING LOCUS T homolog than under short days.
Abstract
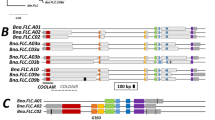
Frequent duplications of the FLOWERING LOCUS T (FT) gene across various taxonomic lineages resulted in FT paralogs with floral repressor function, whereas others duplicates maintained their floral-promoting role. The FT gene has been confirmed as the inducer of photoperiodic flowering in most angiosperms analyzed to date. We identified all FT homologs in the transcriptome of Chenopodium ficifolium and in the genome of Chenopodium suecicum, which are closely related to diploid progenitors of the tetraploid crop Chenopodium quinoa, and estimated their expression during photoperiodic floral induction. We found that expression of FLOWERING LOCUS T like 1 (FTL1), the ortholog of the sugar beet floral activator BvFT2, correlated with floral induction in C. suecicum and short-day C. ficifolium, but not with floral induction in C. ficifolium with accelerated flowering under long days. This C. ficifolium accession was induced to flowering without the concomitant upregulation of any FT homolog.







Similar content being viewed by others
Abbreviations
- AP1 :
-
APETALA 1
- FUL :
-
FRUITFULL
- FT :
-
FLOWERING LOCUS T
- FTL :
-
FLOWERING LOCUS T like
- LFY :
-
LEAFY
References
Ahn JH, Miller D, Winter VJ, Banfield MJ, Jeong HL, So YY, Henz SR, Brady RL, Weigel D (2006) A divergent external loop confers antagonistic activity on floral regulators FT and TFL1. EMBO J 25:605–614. https://doi.org/10.1038/sj.emboj.7600950
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Cao K, Cui L, Zhou X, Ye L, Zou Z, Deng S (2016) Four tomato FLOWERING LOCUS T-Like proteins act antagonistically to regulate floral initiation. Front Plant Sci 6:1213. https://doi.org/10.3389/fpls.2015.01213
Cháb D, Kolář J, Olson MS, Štorchová H (2008) Two Flowering Locus T (FT) homologs in Chenopodium rubrum differ in expression patterns. Planta 228:929–940. https://doi.org/10.1007/s00425-008-0792-3
Drabešová J, Cháb D, Kolař J, Haškovcová K, Štorchová H (2014) A dark-light transition triggers expression of the floral promoter CrFTL1 and downregulates CONSTANS-like genes in a short-day plant Chenopodium rubrum. J Exp Bot 65:2137–2146. https://doi.org/10.1093/jxb/eru073
Drabešová J, Černá L, Mašterová H, Koloušková P, Potocký M, Štorchová H (2016) The Evolution of the FT/TFL1 genes in Amaranthaceae and their expression patterns in the course of vegetative growth and flowering in Chenopodium rubrum. G3-Genes Genomes. Genet 6:3066–3076. https://doi.org/10.1534/g3.116.028639
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797. https://doi.org/10.1093/nar/gkh340
Eriksson S (2006) GA4 is the active gibberellin in the regulation of LEAFY transcription and Arabidopsis floral initiation. Plant Cell 18:2172–2181. https://doi.org/10.1105/tpc.106.042317
Fan C, Hu R, Zhang X, Wang X, Zhang W, Zhang Q, Ma J, Fu YF (2014) Conserved CO-FT regulons contribute to the photoperiod flowering control in soybean. BMC Plant Biol 14:9. https://doi.org/10.1186/1471-2229-14-9
Fuller HJ (1949) Photoperiodic responses of Chenopodium quinoa Willd. and Amaranthus caudatus L. Am J Bot 36:175–180
Goslin K, Zheng B, Serrano-Mislata A et al (2017) Transcription factor interplay between LEAFY and APETALA1/CAULIFLOWER during floral initiation. Plant Physiol 174:1097–1109. https://doi.org/10.1104/pp.17.00098
Grabherr MG, Haas BJ, Yassour M et al (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29:644–652. https://doi.org/10.1038/nbt.1883
Harig L, Beinecke FA, Oltmanns J, Muth J, Müller O, Rüping B, Twyman RM, Fischer R, Prüfer D, Noll GA (2012) Proteins from the FLOWERING LOCUS T-like subclade of the PEBP family act antagonistically to regulate floral initiation in tobacco. Plant J 72:908–921. https://doi.org/10.1111/j.1365-313X.2012.05125.x
Hisamatsu T, King RW (2008) The nature of floral signals in Arabidopsis. II. Roles for FLOWERING LOCUS T (FT) and gibberellin. J Exp Bot 59:3821–3829. https://doi.org/10.1093/jxb/ern232
Ho WWH, Weigel D (2014) Structural features determining flower-promoting activity of Arabidopsis FLOWERING LOCUS T. Plant Cell 26:552–564. https://doi.org/10.1105/tpc.113.115220
Jarvis DE, Ho YS, Lightfoot DJ et al (2017) The genome of Chenopodium quinoa. Nature 542:307–312. https://doi.org/10.1038/nature21370
Kardailsky I, Shukla V, Ahn JH, Dagenais N, Christensen SK, Nguyen JT, Chory J, Harrison MJ, Weigel D (1999) Activation tagging of the floral inducer FT. Science 286:1962–1965. https://doi.org/10.1126/science.286.5446.1962
Kaufmann K, Wellmer F, Muiño JM et al (2010) Orchestration of floral initiation by APETALA1. Science 328:1962–1965. https://doi.org/10.1126/science.1185244
Koskela EA, Mouhu K, Albani MC, Kurokura T, Rantanen M, Sargent DJ, Battey NH, Coupland G, Elomaa P, Hytonen T (2012) Mutation in TERMINAL FLOWER1 reverses the photoperiodic requirement for flowering in the wild strawberry Fragaria vesca. Plant Physiol 159:85–89. https://doi.org/10.1104/pp.112.196659
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359. https://doi.org/10.1038/nmeth.1923
Lee R, Baldwin S, Kenel F, McCallum J, Macknight R (2013) FLOWERING LOCUS T genes control onion bulb formation and flowering. Nat Commun 4:2884. https://doi.org/10.1038/ncomms3884
Li B, Dewey C (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform 12:323. https://doi.org/10.1186/1471-2105-12-323
Li W, Godzik A (2006) Cd-hit: a fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinform 22:1658–1659. https://doi.org/10.1093/bioinformatics/btl158
Liu W, Jiang B, Ma L et al (2018) Functional diversification of Flowering Locus T homologs in soybean: GmFT1a and GmFT2a/5a have opposite roles in controlling flowering and maturation. New Phytol 217:1335–1345. https://doi.org/10.1111/nph.14884
Mandák B, Krak K, Vít P, Pavlíková Z, Lomonosova MN, Habibi F, Lei W, Jellen EN, Douda J (2016) How genome size variation is linked with evolution within Chenopodium sensu lato. Perspect Plant Ecol Evol Syst 23:18–32. https://doi.org/10.1016/j.ppees.2016.09.004
Mandák B, Krak K, Vít P, Lomonosova MN, Belyayev A, Habibi F, Wang L, Douda J, Štorchová H (2018) Hybridization and polyploidization within the Chenopodium album aggregate analysed by means of cytological and molecular markers. Mol Phylogenet Evol 129:189–201. https://doi.org/10.1016/j.ympev.2018.08.016
Mandel AM, Gustafson-Brown C, Savidge B, Yanofsky MF (1992) Molecular characterization of the Arabidopsis floral homeotic gene APETALA1. Nature 360:273–277. https://doi.org/10.1038/360273a0
Matsoukas IG, Massiah AJ, Thomas B (2012) Florigenic and antiflorigenic signaling in plants. Plant Cell Physiol 53:1827–1842. https://doi.org/10.1093/pcp/pcs130
Navarro C, Abelenda JA, Cruz-Oró E, Cuéllar CA, Tamaki S, Silva J, Shimamoto K, Prat S (2011) Control of flowering and storage organ formation in potato by FLOWERING LOCUS T. Nature 478:119–122. https://doi.org/10.1038/nature10431
Pin PA, Benlloch R, Bonnet D, Wremerth-Weich E, Kraft T, Gielen JJL, Nilsson O (2010) An antagonistic pair of FT homologs mediates the control of flowering time in sugar beet. Science 330:1397–1400. https://doi.org/10.1126/science.1197004
Pin PA, Nilsson O (2012) The multifaceted roles of FLOWERING LOCUS T in plant development. Plant Cell Environ 35:1742–1755. https://doi.org/10.1111/j.1365-3040.2012.02558.x
Porri A, Torti S, Romera-Branchat M, Coupland G (2012) Spatially distinct regulatory roles for gibberellins in the promotion of flowering of Arabidopsis under long photoperiods. Development 139:2198–2209. https://doi.org/10.1242/dev.077164
Putterill J, Robson F, Lee K, Simon R, Coupland G (1995) The CONSTANS gene of arabidopsis promotes flowering and encodes a protein showing similarities to zinc finger transcription factors. Cell 80:847–857. https://doi.org/10.1016/0092-8674(95)90288-0
Sather DN, Golenberg EM (2009) Duplication of AP1 within the Spinacia oleracea L. AP1/FUL clade is followed by rapid amino acid and regulatory evolution. Planta 229:507–521. https://doi.org/10.1007/s00425-008-0851-9
Shi J, Dong A, Shen W-H (2015) Epigenetic regulation of rice flowering and reproduction. Front Plant Sci 5:803. https://doi.org/10.3389/fpls.2014.00803
Stamatakis A (2014) RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 30:1312–1313. https://doi.org/10.1093/bioinformatics/btu033
Štorchová H, Hrdličková R, Chrtek J, Tetera M, Fitze D, Fehrer J (2000) An improved method of DNA isolation from plants collected in the field and conserved in saturated NaCl/CTAB solution. Taxon 49:79–84. https://doi.org/10.2307/1223934
Štorchová H, Drabešová J, Cháb D, Kolář J, Jellen EN (2015) The introns in FLOWERING LOCUS T-LIKE (FTL) genes are useful markers for tracking paternity in tetraploid Chenopodium quinoa Willd. Genet Resour Crop Evol 62:913–925. https://doi.org/10.1007/s10722-014-0200-8
Suarez-Lopez P, Wheatley K, Robson F, Onouchi H, Valverde F, Coupland G (2001) CONSTANS mediates between the circadian clock and the control of flowering in Arabidopsis. Nature 410:1116–1120. https://doi.org/10.1038/35074138
Trapnell C, Roberts A, Goff L, Pertea G, Kim D, Kelley DR, Pimentel H, Salzberg SL, Rinn JL, Pachter L (2012) Differential gene and transcript expression analysis of RNA-seq experiments with TopHat and Cufflinks. Nat Protoc 7:562–578. https://doi.org/10.1038/nprot.2012.016
Valverde F (2011) CONSTANS and the evolutionary origin of photoperiodic timing of flowering. J Exp Bot 62:2453–2463. https://doi.org/10.1093/jxb/erq449
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, Van Roy A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3(0034):1. https://doi.org/10.1186/gb-2002-3-7-research0034
Walsh BM, Adhikary D, Maughan PJ, Emshwiller E, Jellen EN (2015) Chenopodium polyploidy inferences from Salt Overly Sensitive 1 (SOS1) data. Am J Bot 102:533–543. https://doi.org/10.3732/ajb.1400344
Weigel D, Alvarez J, Smyth DR, Yanofsky MF, Meyerowitz EM (1992) LEAFY controls floral meristem identity in Arabidopsis. Cell 69:843–859. https://doi.org/10.1016/0092-8674(92)90295-N
Weller JL, Ortega R (2015) Genetic control of flowering time in legumes. Front Plant Sci 6:207. https://doi.org/10.3389/fpls.2015.00207
Wigge PA, Kim MC, Jaeger KE, Busch W, Schmid M, Lohmann JU, Weigel D (2005) Integration of spatial and temporal information during floral induction in Arabidopsis. Science 309:1056–1059. https://doi.org/10.1126/science.1114358
Yang Z (2007) PAML 4: phylogenetic analysis by maximum likelihood. Mol Biol Evol 24:1586–1591. https://doi.org/10.1093/molbev/msm088
Acknowledgements
We would like to thank James D. Stone for the linguistic revision of the text. We are grateful to Lucie Černá for technical assistance in the laboratory and in the greenhouse, and to Marie Pažoutová for bioinformatic aid. We appreciate the helpful comments of the two anonymous reviewers. Our work was supported by the Czech Science Foundation (Grant Number 13-02290S) to BM and HŠ. Additional support was provided by European Regional Development Fund-Project “Centre for Experimental Plant Biology” (No. CZ.02.1.01/0.0/0.0/16_019/0000738) and by long-term research development project RVO 67985939.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Štorchová, H., Hubáčková, H., Abeyawardana, O.A.J. et al. Chenopodium ficifolium flowers under long days without upregulation of FLOWERING LOCUS T (FT) homologs. Planta 250, 2111–2125 (2019). https://doi.org/10.1007/s00425-019-03285-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-019-03285-1