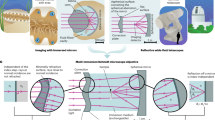
Abstract
The main benefits of the stereomicroscope are that it is designed in a modular motif, allowing for a wide range of accessories such as stands, eyepieces, objectives, and illuminating bases for a wide variety of contrast enhancement techniques. Often utilized to study the surfaces of specimens, the stereomicroscope frequently uses incident (reflected) illumination, permitting the observation of specimens that would normally be too thick or opaque. Translucent and transparent objects can be successfully imaged with a number of transmitted illumination methods depending on the observer’s needs, and fluorescence stereomicroscopes are being increasingly used for three-dimensional observation. An excellent working distance, ranging from 3 to 5 cm to even 20 cm in certain models, and wide field of view that these models feature, are critical factors in the observation of a far-ranging variety of biological specimens. Examples of nervous and other tissues are presented in this review, for example, retina in living fish, paraneurons in shrimp, innervation of murine heart, and YFP-expressing regenerating nerves in the cornea of transgenic mice.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
Change history
24 September 2021
The online version of this book had multiple errors as specified below:
Notes
- 1.
CZ.02.1.01/0.0/0.0/16_019/0000729
Abbreviations
- CMO:
-
Common main objective
- f-number:
-
A parameter inversely proportional to NA of the objective
- FN:
-
Field number (of the eyepiece)
- NA:
-
Numerical aperture
- YFP:
-
Yellow fluorescent protein
References
Bradbury S (1967) The evolution of the microscope. Pergamon Press, New York. ISBN: 9781483164328
Wheatstone C (1838) Contributions to the physiology of vision – part the first. On some remarkable, and hitherto unobserved, phenomena of binocular vision. Phil Trans Roy Soc Lond 128(1838):371–394. https://www.jstor.org/stable/108203
Turner GLE (1989) The great age of the microscope. The collection of the Royal Microscopical Society through 150 years. Adam Hilger, Bristol; CRC Press, New York. ISBN: 9780852740200
Seidenberg RL (1981) Stereomicroscopy: a review. Am Lab 13(April):114–125
Nothnagle PE, Rosenberger HE, Seidenberg RL (1973) Stereomicroscopy: some considerations. Canadian Res Dev 6(2):30–35
Stereomicroscopy (as part of “MicroscopyU: The source for microscopy education”). Molecular Expressions™ (National High Magnetic Field Laboratory, Florida State University, Tallahassee, FL, USA). https://www.microscopyu.com/techniques/stereomicroscopy
Schlueter GE, Gumpertz WE (1976) The stereomicroscope: instrumentation and techniques. Am Lab 8(April):61–71
Mann A (ed) (1993) Selected papers on zoom lenses. SPIE Milestone Series (Thompson BJ, ed), volume MS 85. SPIE, Bellingham, WA. ISBN: 9780819413888
Duval MG, Chung H, Lehmann OJ, Allison WT (2013) Longitudinal fluorescent observation of retinal degeneration and regeneration in zebrafish using fundus lens imaging. Mol Vis 19:1082–1095. https://www.ncbi.nlm.nih.gov/pubmed/23734077
Inoué S, Spring KR (1997) Video microscopy: the fundamentals. Plenum Press, New York. ISBN: 978-0306455315
Schnitzler H, Zimmer K-P (2008) Advances in stereomicroscopy. Proc SPIE 7100:71000P. https://doi.org/10.1117/12.797409
Castinel A, Duignan PJ, Pomroy WE, Lyons ET, Nadler SA, Dailey MD, Wilkinson IS, Chilvers BL (2006) First report and characterization of adult Uncinaria spp. in New Zealand sea lion (Phocarctos hookeri) pups from the Auckland Islands, New Zealand. Parasitol Res 98(4):304–309. https://doi.org/10.1007/s00436-005-0069-8
Pauza DH, Saburkina I, Rysevaite K, Inokaitis H, Jokubauskas M, Jalife J, Pauziene N (2013) Neuroanatomy of the murine cardiac conduction system. A combined stereomicroscopic and fluorescence immunohistochemical study. Auton Neurosci 172(1-2):32–47. https://doi.org/10.1016/j.autneu.2013.01.006
Namavari A, Chaudhary S, Sarkar J, Yco L, Patel K, Han KY, Yue BY, Chang J-H, Jain S (2011) In vivo serial imaging of regenerating corneal nerves after surgical transection in transgenic thy1-YFP mice. Invest Ophthalmol Vis Sci 52(11):8025–8032. https://doi.org/10.1167/iovs.11-8332
Santos VT (2013) Macrobrachium (ghost) shrimp eye. Nikon Small World. https://www.nikonsmallworld.com/people/vitoria-tobias-santos
Murphy DB, Davidson MW (2012) Fundamentals of light microscopy and electronic imaging. Wiley-Blackwell, Hoboken, NJ. https://doi.org/10.1002/9781118382905
Abramowitz M (1990) Reflected light microscopy: an overview. Olympus America, Lehigh Valley, PA. Melville, NY
Abramowitz M (1987) Contrast methods in microscopy: transmitted light. Olympus America, Lehigh Valley, PA. Melville, NY
Axelrod D (1981) Zero-cost modification of bright field microscopes for imaging phase gradient on cells – schlieren optics. Cell Biophys 3(2):167–173. https://doi.org/10.1007/BF02788132
Bradbury S, Evennett PJ (1996) Contrast techniques in light microscopy. RMS Handbooks, № 34. BIOS Scientific Publishers, Oxford. https://doi.org/10.1046/j.1365-2818.1997.00702.x
Tuchin V (2015) Tissue optics: light scattering methods and instruments for medical diagnostics (3rd edn). SPIE, Bellingham, WA. https://doi.org/10.1117/3.1003040
Barer R, Joseph S (1954) Refractometry of living cells, Part I. Basic principles. Quart J Microsci Sci 95(4):399–423. J Cell Sci s3–95:399–423, https://jcs.biologists.org/content/s3-95/32/399
Schlueter GE (1974) A high intensity transmitted light base for darkfield stereomicroscopy. American Laboratory 6(April):40–45
Shaner NC, Patterson GH, Davidson MW (2007) Advances in fluorescent protein technology. J Cell Sci 120(24):4247–4260. https://doi.org/10.1242/jcs.005801
Acknowledgments
The first four coauthors are grateful to the late professor Michael Wesley Davidson (1950–2015). The present chapter is largely derived from the MicroscopyU(niversity) website [6] that was his brainchild. RP acknowledges support via Ministry of Education projects: Chiral Microscopy (LTC17012), CzechBioImaging (LM2015062), and ChemBioDrug.Footnote 1
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Science+Business Media LLC
About this protocol
Cite this protocol
Wilson, E.E., Chambers, W., Pelc, R., Nothnagle, P., Davidson, M.W. (2020). Stereomicroscopy in Neuroanatomy. In: Pelc, R., Walz, W., Doucette, J.R. (eds) Neurohistology and Imaging Techniques. Neuromethods, vol 153. Humana, New York, NY. https://doi.org/10.1007/978-1-0716-0428-1_9
Download citation
DOI: https://doi.org/10.1007/978-1-0716-0428-1_9
Published:
Publisher Name: Humana, New York, NY
Print ISBN: 978-1-0716-0426-7
Online ISBN: 978-1-0716-0428-1
eBook Packages: Springer Protocols