Abstract
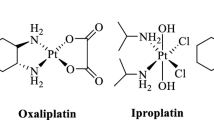
The search for more effective platinum anticancer drugs has led to the design, synthesis, and preclinical testing of hundreds of new platinum complexes. This search resulted in the recognition and subsequent FDA approval of the third-generation Pt(II) anticancer drug, [Pt(1,2-diaminocyclohexane)(oxalate)], oxaliplatin, as an effective agent in treating colorectal and gastrointestinal cancers. Another promising example of the class of anticancer platinum(II) complexes incorporating the Pt(1,n-diaminocycloalkane) moiety is kiteplatin ([Pt(cis-1,4-DACH)Cl2], DACH = diaminocyclohexane). We report here our progress in evaluating the role of the cycloalkyl moiety in these complexes focusing on the synthesis, characterization, evaluation of the antiproliferative activity in tumor cells and studies of the mechanism of action of new [Pt(cis-1,3-diaminocycloalkane)Cl2] complexes wherein the cis-1,3-diaminocycloalkane group contains the cyclobutyl, cyclopentyl, and cyclohexyl moieties. We demonstrate that [Pt(cis-1,3-DACH)Cl2] destroys cancer cells with greater efficacy than the other two investigated 1,3-diamminocycloalkane derivatives, or cisplatin. Moreover, the investigated [Pt(cis-1,3-diaminocycloalkane)Cl2] complexes show selectivity toward tumor cells relative to non-tumorigenic normal cells. We also performed several mechanistic studies in cell-free media focused on understanding some early steps in the mechanism of antitumor activity of bifunctional platinum(II) complexes. Our data indicate that reactivities of the investigated [Pt(cis-1,3-diaminocycloalkane)Cl2] complexes and cisplatin with glutathione and DNA binding do not correlate with antiproliferative activity of these platinum(II) complexes in cancer cells. In contrast, we show that the higher antiproliferative activity in cancer cells of [Pt(cis-1,3-DACH)Cl2] originates from its highest hydrophobicity and most efficient cellular uptake.
Graphic abstract





Similar content being viewed by others
References
Connors TA, Jones M, Ross WCJ, Braddock PD, Khokhar AR, Tobe ML (1972) New platinum complexes with anti-tumour activity. Chem Biol Interact 5:415–424
Cleare MJ, Hoeschele JD (1973) Antitumor activity of group viii transition metal complexes. 1. Platinum(ll) complexes. Bioinorg Chem 2:187–210
Gale GR, Walker EM Jr, Atkins LM, Smith AB, Meischen SJ (1974) Antileukemic properties of dichloro(1,2-diaminocyclohexane)platinum(II). Res Commun Chem Pathol Pharmacol 7:529–538
Kidani Y, Inagaki K, Iigo M, Hoshi A, Kuretani K (1978) Antitumor activity of 1,2-diamminocyclohexane-platinum complexes against sarcoma 180 ascites form. J Med Chem 21:1315–1318
Kim GP, Erlichman C (2007) Oxaliplatin in the treatment of colorectal cancer. Exp Opin Drug Metabol Toxicol 3:281–294
Kochi M, Fujii M, Kanamori N, Kaiga T, Okubo R, Mihara Y, Takayama T (2011) Pharmacokinetics of oxaliplatin in gastrointestinal cancer patients with malignant ascites. J Chemother 23:28–31
Hoeschele JD, Petruzzella E, Margiotta N, Natile G, Gandin V, Marzano C (2014) Kiteplatin—a potential clinical candidate showing significant activity vs Oxaliplatin-resistant colorectal cancer. Anticancer Res 34:5949–5950
Johnstone TC, Suntharalingam K, Lippard SJ (2016) The next generation of platinum drugs: Targeted pt(II) agents, nanoparticle delivery, and pt(iv) prodrugs. Chem Rev 116:3436–3486
Brabec V, Hrabina O, Kasparkova J (2017) Cytotoxic platinum coordination compounds DNA binding agents. Coord Chem Rev 351:2–31
Dhara SC (1970) Process for the production of pure cis-platinum-(II)-diammine dichloride. Indian J Chem 8:193–194
Geyer R, Nordemann U, Strasser A, Wittmann H-J, Buschauer A (2016) Conformational restriction and enantioseparation increase potency and selectivity of cyanoguanidine-type histamine h4 receptor agonists. J Med Chem 59:3452–3470
Sheldrick G (2015) Shelxt—integrated space-group and crystal-structure determination. Acta Crystallog Sect. A 71:3–8
Dolomanov OV, Bourhis LJ, Gildea RJ, Howard JAK, Puschmann H (2009) Olex2: A complete structure solution, refinement and analysis program. J Appl Crystallog 42:339–341
Sheldrick GM (2008) A short history of shelx. Acta Crystallogr Sect A 64:112–122
Kasparkova J, Kostrhunova H, Novohradsky V, Pracharova J, Curci A, Margiotta N, Natile G, Brabec V (2017) Anticancer kiteplatin pyrophosphate derivatives show unexpected target selectivity for DNA. Dalton Trans 46:14139–14148
Hreusova M, Novákova O, Kostrhunova H, Pracharova J, Brabec V, Kašpárková J (2019) DNA modification by cisplatin-like Pt(II) complexes containing 1,1′-binaphtyl-2,2′-diamine ligand does not correlate with their antiproliferative activity in cancer cells. Inorg Chim Acta 495:118952
Novakova O, Liskova B, Vystrcilova J, Suchankova T, Vrana O, Starha P, Travnicek Z, Brabec V (2014) Conformation and recognition of DNA damaged by antitumor cis-dichlorido platinum(ii) complex of cdk inhibitor bohemine. Eur J Med Chem 78:54–64
Dabrowiak JC, Goodisman J, Souid AK (2002) Kinetic study of the reaction of cisplatin with thiols. Drug Metab Dispos 30:1378–1384
Qu Y, Wenxia T, Hu H, Dai A, Ji X, Zhang F and Liu Y (1986) Synthesis,characterization and antitumour activity of platinum complexes of 1,3-diaminocyclohexane. Chinese J Appl Chem 3:25–29
Kasparkova J, Suchankova T, Halamikova A, Zerzankova L, Vrana O, Margiotta N, Natile G, Brabec V (2010) Cytotoxicity, cellular uptake, glutathione and DNA interactions of an antitumor large-ring ptii chelate complex incorporating the cis-1,4-diaminocyclohexane carrier ligand. Biochem Pharmacol 79:552–564
Margiotta N, Savino S, Marzano C, Pacifico C, Hoeschele JD, Gandin V, Natile G (2016) Cytotoxicity-boosting of kiteplatin by Pt(IV) prodrugs with axial benzoate ligands. J Inorg Biochem 160:85–93
Kostrhunova H, Zajac J, Novohradsky V, Kasparkova J, Malina J, Aldrich-Wright JR, Petruzzella E, Sirota R, Gibson D, Brabec V (2019) A subset of new platinum antitumor agents kills cells by a multimodal mechanism of action also involving changes in the organization of the microtubule cytoskeleton. J Med Chem 62:5176–5190
Margiotta N, Marzano C, Gandin V, Osella D, Ravera M, Gabano E, Platts JA, Petruzzella E, Hoeschele JD, Natile G (2012) Revisiting [PtCl2(cis-1,4-dach)]: An underestimated antitumor drug with potential application to the treatment of oxaliplatin-refractory colorectal cancer. J Med Chem 55:7182–7192
Wang D, Lippard SJ (2005) Cellular processing of platinum anticancer drugs. Nat Rev Drug Discov 4:307–320
Novohradsky V, Liu Z, Vojtiskova M, Sadler PJ, Brabec V, Kasparkova J (2014) Mechanism of cellular accumulation of an iridium(iii) pentamethylcyclopentadienyl anticancer complex containing a C, N-chelating ligand. Metallomics 6:682–690
Safaei R, Howell SB (2005) Copper transporters regulate the cellular pharmacology and sensitivity to pt drugs. Crit Rev Oncol Hematol 53:13–23
Reedijk J (1999) Why does cisplatin reach guanine-N7 with competing s-donor ligands avaiable in the cell? Chem Rev 99:2499–2510
Hagrman D, Goodisman J, Dabrowiak JC, Souid AK (2003) Kinetic study on the reaction of cisplatin with metallothionein. Drug Metab Disp 31:916–923
Johnson NP, Butour J-L, Villani G, Wimmer FL, Defais M, Pierson V, Brabec V (1989) Metal antitumor compounds: the mechanism of action of platinum complexes. Prog Clin Biochem Med 10:1–24
Johnstone TC, Alexander SM, Lin W, Lippard SJ (2014) Effects of monofunctional platinum agents on bacterial growth: a retrospective study. J Am Chem Soc 136:116–118
Kasparkova J, Brabec V (2015) Effects of antitumor derivatives of ineffective transplatin on bacterial cells: is DNA a pharmacological target? J Inorg Biochem 153:206–210
Acknowledgements
This work was supported by the Ministry of Education of the Czech Republic [Grant Number LTAUSA18009]. The authors acknowledge that Richard Staples from the Chemistry Department, Michigan State University determined the crystal structures of our three Pt(cis-1,3-diaminocycloalkane)Cl2 samples and Dan Holmes from the Chemistry Department, Michigan State University obtained the 1H and 195Pt NMR spectral data for these complexes.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
The structural data are deposited with the Cambridge Crystallographic Data Center: 2021147 ([Pt(cis-1,3-diaminocyclobutane)Cl2]), 2021148 ([Pt(cis-1,3-diaminocyclopentane)Cl2]), 2021149 ([Pt(cis-1,3-diaminocyclohexane)Cl2]).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hoeschele, J.D., Kasparkova, J., Kostrhunova, H. et al. Synthesis, antiproliferative activity in cancer cells and DNA interaction studies of [Pt(cis-1,3-diaminocycloalkane)Cl2] analogs. J Biol Inorg Chem 25, 913–924 (2020). https://doi.org/10.1007/s00775-020-01809-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-020-01809-9