Abstract
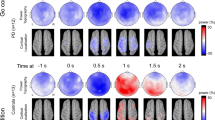
Although deep brain stimulation of the subthalamic nucleus (STN-DBS) in Parkinson’s disease (PD) is generally a successful therapy, adverse events and insufficient clinical effect can complicate the treatment in some patients. We studied clinical parameters and cortical oscillations related to STN-DBS to identify patients with suboptimal responses. High-density EEG was recorded during a visual oddball three-stimuli paradigm in DBS “off” and “on” conditions in 32 PD patients with STN-DBS. Pre-processed data were reconstructed into the source space and the time–frequency analysis was evaluated. We identified a subgroup of six patients with longer reaction times (RT) during the DBS “on” state than in the DBS “off” state after target stimuli. These subjects had lower motor responsiveness to DBS and decreased memory test results compared to the other subjects. Moreover, the alpha and beta power decrease (event-related desynchronizations, ERD), known as an activation correlate linked to motor and cognitive processing, was also reduced in the DBS “on” condition in these patients. A subgroup of PD patients with a suboptimal response to STN-DBS was identified. Evaluation of RT could potentially serve as a biomarker for responsiveness to STN-DBS.





Similar content being viewed by others
References
Androulidakis AG, Kühn AA, Chen CC, Blomstedt P, Kempf F, Kupsch A et al (2007) Dopaminergic therapy promotes lateralized motor activity in the subthalamic area in Parkinson’s disease. Brain 130(Pt 2):457–468. https://doi.org/10.1093/brain/awl358
Balestrino R, Baroncini D, Fichera M, Donofrio CA, Franzin A, Mortini P et al (2017) Weight gain after subthalamic nucleus deep brain stimulation in Parkinson’s disease is influenced by dyskinesias’ reduction and electrodes’ position. Neurol Sci 38(12):2123–2129. https://doi.org/10.1007/s10072-017-3102-7
Benabid AL, Chabardes S, Mitrofanis J, Pollak P (2009) Deep brain stimulation of the subthalamic nucleus for the treatment of Parkinson’s disease. Lancet Neurol 8(1):67–81. https://doi.org/10.1016/S1474-4422(08)70291-6
Bočková M, Rektor I (2019) Impairment of brain functions in Parkinson’s disease reflected by alterations in neural connectivity in EEG studies: a viewpoint. Clin Neurophysiol 130(2):239–247. https://doi.org/10.1016/j.clinph.2018.11.013
Bočková M, Chládek J, Šímová L, Jurák P, Halámek J, Rektor I (2013) Oscillatory changes in cognitive networks activated during a three-stimulus visual paradigm: an intracerebral study. Clin Neurophysiol 124(2):283–291. https://doi.org/10.1016/j.clinph.2012.07.009
Bonanni L, Thomas A, Tiraboschi P, Perfetti B, Varanese S, Onofrj M (2008) EEG comparisons in early Alzheimer’s disease, dementia with Lewy bodies and Parkinson’s disease with dementia patients with a 2-year follow-up. Brain 131(Pt 3):690–705. https://doi.org/10.1093/brain/awm322
Brown P (2003) Oscillatory nature of human basal ganglia activity: relationship to the pathophysiology of Parkinson’s disease. Mov Disord 18(4):357–363. https://doi.org/10.1002/mds.10358
Brown P (2006) Bad oscillations in Parkinson’s disease. J Neural Transm Suppl 70:27–30. https://doi.org/10.1007/978-3-211-45295-0_6
Chen CC, Hsu YT, Chan HL, Chiou SM, Tu PH, Lee ST et al (2010) Complexity of subthalamic 13–35 Hz oscillatory activity directly correlates with clinical impairment in patients with Parkinson’s disease. Exp Neurol 224(1):234–240. https://doi.org/10.1016/j.expneurol.2010.03.015
Christ BU, Combrinck MI, Thomas KGF (2018) Both reaction time and accuracy measures of intraindividual variability predict cognitive performance in Alzheimer’s disease. Front Hum Neurosci 12:124
Coito A, Michel CM, van Mierlo P, Vulliemoz S, Plomp G (2016) Directed functional brain connectivity based on EEG source imaging: methodology and application to temporal lobe epilepsy. IEEE Trans Biomed Eng 63(12):2619–2628. https://doi.org/10.1109/TBME.2016.2619665
Conroy MA, Polich J (2007) Normative variation of P3a and P3b from a large sample (N = 120): gender, topography, and response time. J Psychophysiol 21:22–32. https://doi.org/10.1027/0269-8803.21.1.22
Cosgrove J, Picardi C, Smith SL, Lones MA, Jamieson S, Alty JE (2016) Investigating the relationship between reaction time and cognition in Parkinson's disease. Mov Disord 31:S458–S458
DeLong MR, Huang KT, Gallis J, Lokhnygina Y, Parente B, Hickey P et al (2014) Effect of advancing age on outcomes of deep brain stimulation for Parkinson disease. JAMA Neurol 71(10):1290–1295
Delorme A, Makeig S (2004) EEGLAB: An open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J Neurosci Methods 134(1):9–21. https://doi.org/10.1016/j.jneumeth.2003.10.009
Ewert S, Plettig P, Li N, Chakravarty MM, Collins D, L, Herrington TM, et al (2018) Toward defining deep brain stimulation targets in MNI space: a subcortical atlas based on multimodal MRI, histology and structural connectivity. Neuroimage 170:271–282
Ferreira D, Jelic V, Cavallin L, Oeksengaard AR, Snaedal J, Høgh P et al (2016) Electroencephalography is a good complement to currently established dementia biomarkers. Dement Geriatr Cogn Disord 42(1–2):80–92. https://doi.org/10.1159/000448394
Fumagalli M, Giannicola G, Rosa M, Marceglia S, Lucchiari C, Mrakic-Sposta S et al (2011) Conflict-dependent dynamic of subthalamic nucleus oscillations during moral decisions. Soc Neurosci 6(3):243–256. https://doi.org/10.1080/17470919.2010.515148
Giannicola G, Marceglia S, Rossi L, Mrakic-Sposta S, Rampini P, Tamma F et al (2010) The effects of levodopa and ongoing deep brain stimulation on subthalamic beta oscillations in Parkinson’s disease. Exp Neurol 226(1):120–127. https://doi.org/10.1016/j.expneurol.2010.08.011
Habets JGV, Heijmans M, Kuijf ML, Janssen MLF, Temel Y, Kubben PL (2018) An update on adaptive deep brain stimulation in Parkinson’s disease. Mov Disord 33(12):1834–1843. https://doi.org/10.1002/mds.115
Hirschmann J, Özkurt TT, Butz M, Homburger M, Elben S, Hartmann CJ et al (2011) Distinct oscillatory STN-cortical loops revealed by simultaneous MEG and local field potential recordings in patients with Parkinson’s disease. Neuroimage 55(3):1159–1168. https://doi.org/10.1016/j.neuroimage.2010.11.063
Horn A, Kühn AA (2015) Lead-DBS: a toolbox for deep brain stimulation electrode localizations and visualizations. Neuroimage 107:127–135
Huebl J, Spitzer B, Brücke C, Schönecker T, Kupsch A, Alesch F et al (2014) Oscillatory subthalamic nucleus activity is modulated by dopamine during emotional processing in Parkinson’s disease. Cortex 60:69–81. https://doi.org/10.1016/j.cortex.2014.02.019
Jordan N, Sagar HJ, Cooper JA (1992) Cognitive components of reaction time in Parkinson’s disease. J Neurol Neurosurg Psychiatry 55(8):658–664
Kojovic M, Mir P, Trender-Gerhard I, Schneider SA, Pareés I, Edwards MJ, Bhatia KP, Jahanshahi M (2014) Motivational modulation of bradykinesia in Parkinson’s disease off and on dopaminergic medication. J Neurol 261(6):1080–1089
Kühn AA, Kupsch A, Schneider GH, Brown P (2006) Reduction in subthalamic 8–35 Hz oscillatory activity correlates with clinical improvement in Parkinson’s disease. Eur J Neurosci 23(7):1956–1960. https://doi.org/10.1111/j.1460-9568.2006.04717.x
Kumru H, Summerfield C, Valldeoriola F, Valls-Solé J (2004) Effects of subthalamic nucleus stimulation on characteristics of EMG activity underlying reaction time in Parkinson’s disease. Mov Disord 19(1):94–100
Litvak V, Eusebio A, Jha A, Oostenveld R, Barnes GR, Penny WD et al (2010) Optimized beamforming for simultaneous MEG and intracranial local field potential recordings in deep brain stimulation patients. Neuroimage 50(4):1578–1588. https://doi.org/10.1016/j.neuroimage.2009.12.115
Litvak V, Jha A, Eusebio A, Oostenveld R, Foltynie T, Limousin P et al (2011) Resting oscillatory cortico-subthalamic connectivity in patients with Parkinson’s disease. Brain 134(Pt 2):359–374. https://doi.org/10.1093/brain/awq332
Lozano AM, Lipsman N, Bergman H, Brown P, Chabardes S, Chang JW et al (2019) Deep brain stimulation: current challenges and future directions. Nat Rev Neurol 15(3):148–160
Moro E, Lozano AM, Pollak P, Agid Y, Rehncrona S, Volkmann J et al (2010) Long-term results of a multicenter study on subthalamic and pallidal stimulation in Parkinson’s disease. Mov Disord 25:578–586
Pfurtscheller G (2001) Functional brain imaging based on ERD/ERS. Vision Res 41(10–11):1257–1260. https://doi.org/10.1016/S0042-6989(00)00235-2
Pinter M, Birk M, Helscher RJ, Binder H (1999) Short-term effect of amantadine sulphate on motor performance and reaction time in patients with idiopathic Parkinson’s disease. J Neural Transm (Vienna) 106(7–8):711–724
Plesinger F, Jurco J, Halamek J, Jurak P (2016) SignalPlant: an open signal processing software platform. Physiol Meas 37(7):N38–48. https://doi.org/10.1088/0967-3334/37/7/N38
Polich J (2007) Updating P300: an integrative theory of P3a and P3b. Clin Neurophysiol 118(10):2128–2148. https://doi.org/10.1016/j.clinph.2007.04.019
Rodriguez-Oroz MC, López-Azcárate J, Garcia-Garcia D, Alegre M, Toledo J, Valencia M et al (2011) Involvement of the subthalamic nucleus in impulse control disorders associated with Parkinson’s disease. Brain 134(Pt 1):36–49. https://doi.org/10.1093/brain/awq301
Rosa M, Fumagalli M, Giannicola G, Marceglia S, Lucchiari C, Servello D et al (2013) Pathological gambling in Parkinson’s disease: subthalamic oscillations during economics decisions. Mov Disord 28(12):1644–1652. https://doi.org/10.1002/mds.25427
Saint-Cyr JA, Trépanier LL, Kumar R, Lozano AM, Lang AE (2000) Neuropsychological consequences of chronic bilateral stimulation of the subthalamic nucleus in Parkinson’s disease. Brain 123(Pt 10):2091–2108. https://doi.org/10.1093/brain/123.10.2091
Schuepbach WMM, Rau J, Knudsen K, Volkmann J, Krack P, Timmermann L et al (2013) Neurostimulation for Parkinson’s disease with early motor complications. N Engl J Med 368(7):610–622
Seeber M, Cantonas LM, Hoevels M, Sesia T, Visser-Vandewalle V, Michel CM (2019) Subcortical electrophysiological activity is detectable with high-density EEG source imaging. Nat Commun 10(1):753. https://doi.org/10.1038/s41467-019-08725-w
Temel Y, Kessels A, Tan S, Topdag A, Boon P, Visser-Vandewalle V (2006) Behavioural changes after bilateral subthalamic stimulation in advanced Parkinson disease: a systematic review. Parkinsonism Relat Disord 12(5):265–272. https://doi.org/10.1016/j.parkreldis.2006.01.004
Witt K, Daniels C, Reiff J, Krack P, Volkmann J, Pinsker MO et al (2008) Neuropsychological and psychiatric changes after deep brain stimulation for Parkinson’s disease: a randomised, multicentre study. Lancet Neurol 7(7):605–614. https://doi.org/10.1016/S1474-4422(08)70114-5
Acknowledgements
Thanks to Anne Johnson for English language assistance.
Funding
This research has been financially supported by grant AZV 16-33798A. The research has been also supported by CEITEC 2020, LQ1601, The Ministry of Education, Youth and Sports of the Czech Republic/MEYS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Ethical approval
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bočková, M., Lamoš, M., Klimeš, P. et al. Suboptimal response to STN-DBS in Parkinson’s disease can be identified via reaction times in a motor cognitive paradigm. J Neural Transm 127, 1579–1588 (2020). https://doi.org/10.1007/s00702-020-02254-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-020-02254-3