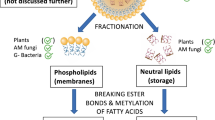
Abstract
Specific biomarker molecules are increasingly being used for detection and quantification in plant and soil samples of arbuscular mycorrhizal (AM) fungi, an important and widespread microbial guild heavily implicated in transfers of nutrients and carbon between plants and soils and in the maintenance of soil physico-chemical properties. Yet, concerns have previously been raised as to the validity of a range of previously used approaches (e.g., microscopy, AM-specific fatty acids, sterols, glomalin-like molecules, ribosomal DNA sequences), justifying further research into novel biomarkers for AM fungal abundance and/or functioning. Here, we focused on complex polar lipids contained in pure biomass of Rhizophagus irregularis and in nonmycorrhizal and mycorrhizal roots of chicory (Cichorium intybus), leek (Allium porrum), and big bluestem (Andropogon gerardii). The lipids were analyzed by shotgun lipidomics using a high-resolution hybrid mass spectrometer. Size range between 1350 and 1550 Da was chosen for the detection of potential biomarkers among cardiolipins (1,3-bis(sn-3′-phosphatidyl)-sn-glycerols), a specific class of phospholipids. The analysis revealed a variety of molecular species, including cardiolipins containing one or two polyunsaturated fatty acids with 20 carbon atoms each, i.e., arachidonic and/or eicosapentaenoic acids, some of them apparently specific for the mycorrhizal samples. Although further verification using a greater variety of AM fungal species and samples from various soils/ecosystems/environmental conditions is needed, current results suggest the possibility to identify novel biochemical signatures specific for AM fungi within mycorrhizal roots. Whether they could be used for quantification of both root and soil colonization by the AM fungi merits further scrutiny.

Similar content being viewed by others
Data availability
All data generated during this study are included in this published article or its electronic supplements.
References
Arruda B, Rodrigues YF, Herrera WFB, Robin A, Cotta SR, Andreote FD (2022) Experimental validation under controlled conditions of real time PCR to quantify arbuscular mycorrhizal colonization in root. J Microbiol Meth 192:106382. https://doi.org/10.1016/j.mimet.2021.106382
Balser TC, Treseder KK, Ekenler M (2005) Using lipid analysis and hyphal length to quantify AM and saprotrophic fungal abundance along a soil chronosequence. Soil Biol Biochem 37:601–604. https://doi.org/10.1016/j.soilbio.2004.08.019
Bardgett RD, van der Putten WH (2014) Belowground biodiversity and ecosystem functioning. Nature 515:505–511. https://doi.org/10.1038/nature13855
Beilby JP, Kidby DK (1980) Biochemistry of ungerminated and germinated spores of the vesicular-arbuscular mycorrhizal fungus, Glomus caledonius: changes in neutral and polar lipids. J Lipid Res 21:739–750. https://doi.org/10.1016/S0022-2275(20)34801-X
Bligh E, Dyer W (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37:911–917. https://doi.org/10.1139/o59-099
Brands M, Cahoon EB, Dörmann P (2020) Palmitvaccenic acid (Δ11-cis-hexadecenoic acid) is synthesized by an OLE1-like desaturase in the arbuscular mycorrhiza fungus Rhizophagus irregularis. Biochemistry 59:1163–1172. https://doi.org/10.1021/acs.biochem.0c00051
Chen W, Cai C, Sheu S (2020) Sandaracinomonas limnophila gen. nov., sp. nov., a new member of the family Cytophagaceae isolated from a freshwater mesocosm. Int J Syst Evol Microbiol 70:2178–2185. https://doi.org/10.1099/ijsem.0.003532
Christie WW (2023) The LipidWeb [WWW Document]. URL https://www.lipidmaps.org/resources/lipidweb/lipidweb_html/index.html
Couillerot O, Ramírez-Trujillo A, Walker V, von Felten A, Jansa J, Maurhofer M, Défago G, Prigent-Combaret C, Comte G, Caballero-Mellado J, Moënne-Loccoz Y (2013) Comparison of prominent Azospirillum strains in Azospirillum–Pseudomonas–Glomus consortia for promotion of maize growth. Appl Microbiol Biotech 97:4639–4649. https://doi.org/10.1007/s00253-012-4249-z
Cronan JE, Thomas J (2009) Bacterial fatty acid synthesis and its relationships with polyketide synthetic pathways. Meth Enzymol 459:395–433. https://doi.org/10.1016/S0076-6879(09)04617-5
Ezeagu IE, Petzke KJ, Lange E, Metges CC (1998) Fat content and fatty acid composition of oils extracted from selected wild-gathered tropical plant seeds from Nigeria. J Amer Oil Chem Soc 75:1031–1035. https://doi.org/10.1007/s11746-998-0282-6
Fakas S, Papanikolaou S, Galiotou-Panayotou M, Komaitis M, Aggelis G (2006) Lipids of Cunninghamella echinulata with emphasis to γ-linolenic acid distribution among lipid classes. Appl Microbiol Biotech 73:676–683. https://doi.org/10.1007/s00253-006-0506-3
Field KJ, Pressel S (2018) Unity in diversity: structural and functional insights into the ancient partnerships between plants and fungi. New Phytol 220:996–1011. https://doi.org/10.1111/nph.15158
Field KJ, Bidartondo MI, Rimington WR, Hoysted GA, Beerling DJ, Cameron DD, Duckett JG, Leake JR, Pressel S (2019) Functional complementarity of ancient plant–fungal mutualisms: contrasting nitrogen, phosphorus and carbon exchanges between Mucoromycotina and Glomeromycotina fungal symbionts of liverworts. New Phytol 223:908–921. https://doi.org/10.1111/nph.15819
Fontaine J, Grandmougin-Ferjani A, Hartmann M, Sancholle M (2001) Sterol biosynthesis by the arbuscular mycorrhizal fungus Glomus intraradices. Lipids 36:1357–1363. https://doi.org/10.1007/s11745-001-0852-z
Frey B, Buser H, Schuepp H (1992) Identification of ergosterol in vesicular-arbuscular mycorrhizae. Biol Fert Soils 13:229–234. https://doi.org/10.1007/BF00340581
Frey B, Vilarino A, Schuepp H, Arines J (1994) Chitin and ergosterol content of extraradical and intraradical mycelium of the vesicular-arbuscular mycorrhizal fungus Glomus intraradices. Soil Biol Biochem 26:711–717. https://doi.org/10.1016/0038-0717(94)90263-1
Frostegård A, Tunlid A, Bååth E (2011) Use and misuse of PLFA measurements in soils. Soil Biol Biochem 43:1621–1625. https://doi.org/10.1016/j.soilbio.2010.11.021
Groenewald EG, van der Westhuizen AJ (1997) Prostaglandins and related substances in plants. Bot Rev 63:199–220. https://doi.org/10.1007/BF02857948
Gu Z, Valianpour F, Chen S, Vaz FM, Hakkaart GA, Wanders RJA, Greenberg ML (2004) Aberrant cardiolipin metabolism in the yeast taz1 mutant: a model for Barth syndrome. Mol Microbiol 51:149–158. https://doi.org/10.1046/j.1365-2958.2003.03802.x
Hanssen J, Thingstad T, Goksoyr J (1974) Evaluation of hyphal lengths and fungal biomass in soil by a membrane-filter technique. Oikos 25:102–107. https://doi.org/10.2307/3543552
Heidarianpour MB, Aliasgharzad N, Olsson PA (2020) Positive effects of co-inoculation with Rhizophagus irregularis and Serendipita indica on tomato growth under saline conditions, and their individual colonization estimated by signature lipids. Mycorrhiza 30:455–466. https://doi.org/10.1007/s00572-020-00962-y
Holátko J, Brtnický M, Kučerík J, Kotianová M, Elbl J, Kintl A, Kynický J, Benada O, Datta R, Jansa J (2021) Glomalin – truths, myths, and the future of this elusive soil glycoprotein. Soil Biol Biochem 153:108116. https://doi.org/10.1016/j.soilbio.2020.108116
Horvath SE, Daum G (2013) Lipids of mitochondria. Prog Lipid Res 52:590–614. https://doi.org/10.1016/j.plipres.2013.07.002
Hoysted GA, Kowal J, Jacob A, Rimington WR, Duckett JG, Pressel S, Orchard S, Ryan MH, Field KJ, Bidartondo MI (2018) A mycorrhizal revolution. Curr Opin Plant Biol 44:1–6. https://doi.org/10.1016/j.pbi.2017.12.004
Islam T, Larsen O, Birkeland N (2020) A novel cold-adapted Methylovulum species, with a high C16:1ω5c content, isolated from an arctic thermal spring in Spitsbergen. Microbes Environ 35:ME20044. https://doi.org/10.1264/jsme2.ME20044
Janoušková M, Püschel D, Hujslová M, Slavíková R, Jansa J (2015) Quantification of arbuscular mycorrhizal fungal DNA in roots: how important is material preservation? Mycorrhiza 25:205–214. https://doi.org/10.1007/s00572-014-0602-7
Joergensen R (2022) Phospholipid fatty acids in soil-drawbacks and future prospects. Biol Fert Soils 58:1–6. https://doi.org/10.1007/s00374-021-01613-w
Joung Y, Seo M, Kang H, Kim H, Ahn T, Cho J, Joh K (2015) Emticicia aquatica sp nov., a species of the family Cytophagaceae isolated from fresh water. Int J Syst Evol Microbiol 65:4358–4362. https://doi.org/10.1099/ijsem.0.000577
Kiers ET, Duhamel M, Beesetty Y, Mensah JA, Franken O, Verbruggen E, Fellbaum CR, Kowalchuk GA, Hart MM, Bago A, Palmer TM, West SA, Vandenkoornhuyse P, Jansa J, Bücking H (2011) Reciprocal rewards stabilize cooperation in the mycorrhizal symbiosis. Science 333:880–882. https://doi.org/10.1126/science.1208473
Konvalinková T, Püschel D, Řezáčová V, Gryndlerová H, Jansa J (2017) Carbon flow from plant to arbuscular mycorrhizal fungi is reduced under phosphorus fertilization. Plant Soil 419:319–333. https://doi.org/10.1007/s11104-017-3350-6
Koske RE, Gemma JN (1989) A modified procedure for staining roots to detect VA mycorrhizas. Mycol Res 92:486–488. https://doi.org/10.1016/S0953-7562(89)80195-9
Kotani S, Izawa S, Komai N, Takayanagi A, Arioka M (2016) Mitochondria-localized phospholipase A2, AoPlaA, in Aspergillus oryzae displays phosphatidylethanolamine-specific activity and is involved in the maintenance of mitochondrial phospholipid composition. Fungal Genet Biol 96:1–11. https://doi.org/10.1016/j.fgb.2016.09.001
Kumari A, Pabbi S (2023) Tyagi A (2023) Recent advances in enhancing the production of long chain omega-3 fatty acids in microalgae. Crit Rev Food Sci Nutr 10(1080/10408398):2226720
Kuyper TW, Jansa J (2023) Arbuscular mycorrhiza: advances and retreats in our understanding of the ecological functioning of the mother of all root symbioses. Plant Soil 489:41–88. https://doi.org/10.1007/s11104-023-06045-z
Lee JC, Yang JS, Moon MH (2019) Simultaneous relative quantification of various polyglycerophospholipids with isotope-labeled methylation by nanoflow ultrahigh performance liquid chromatography-tandem mass spectrometry. Anal Chem 91:6716–6723. https://doi.org/10.1021/acs.analchem.9b00800
Lekberg Y, Bååth E, Frostegård Å, Hammer E, Hedlund K, Jansa J, Kaiser C, Ramsey PW, Řezanka T, Rousk J, Wallander H, Welc M, Olsson PA (2022) Fatty acid 16:1ω5 as a proxy for arbuscular mycorrhizal fungal biomass: current challenges and ways forward. Biol Fert Soils 58:835–842. https://doi.org/10.1007/s00374-022-01670-9
Lu H, Chen H, Tang X, Yang Q, Zhang H, Chen YQ, Chen W (2019) Ultra performance liquid chromatography–Q exactive orbitrap/mass spectrometry-based lipidomics reveals the influence of nitrogen sources on lipid biosynthesis of Mortierella alpina. J Agr Food Chem 67:10984–10993. https://doi.org/10.1021/acs.jafc.9b04455
Lu Y, Eiriksson FF, Thorsteinsdóttir M, Cronberg N, Simonsen HT (2023) Lipidomes of Icelandic bryophytes and screening of high contents of polyunsaturated fatty acids by using lipidomics approach. Phytochemistry 206:113560. https://doi.org/10.1016/j.phytochem.2022.113560
Matthäus B (2012) The database Seed Oil Fatty Acids (SOFA) is back on the Internet! Eur J Lipid Sci Tech 114:701–702. https://doi.org/10.1002/ejlt.201200160
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular—arbuscular mycorrhizal fungi. New Phytol 115:495–501. https://doi.org/10.1111/j.1469-8137.1990.tb00476.x
Nichols P, Stulp B, Jones J, White D (1986) Comparison of fatty-acid content and DNA homology of the filamentous gliding bacteria Vitreoscilla, Flexibacter, Filibacter. Arch Microbiol 146:1–6. https://doi.org/10.1007/BF00690149
Ohlrogge J, Thrower N, Mhaske V, Stymne S, Baxter M, Yang W, Liu J, Shaw K, Shorrosh B, Zhang M, Wilkerson C, Matthäus B (2018) PlantFAdb: a resource for exploring hundreds of plant fatty acid structures synthesized by thousands of plants and their phylogenetic relationships. Plant J 96:1299–1308. https://doi.org/10.1111/tpj.14102
Olsson P (1999) Signature fatty acids provide tools for determination of the distribution and interactions of mycorrhizal fungi in soil. FEMS Microbiol Ecol 29:303–310. https://doi.org/10.1016/S0168-6496(99)00021-5
Olsson PA, Lekberg Y (2022) A critical review of the use of lipid signature molecules for the quantification of arbuscular mycorrhiza fungi. Soil Biol Biochem 166:108574. https://doi.org/10.1016/j.soilbio.2022.108574
Olsson P, Larsson L, Bago B, Wallander H, van Aarle I (2003) Ergosterol and fatty acids for biomass estimation of mycorrhizal fungi. New Phytol 159:7–10. https://doi.org/10.1046/j.1469-8137.2003.00810.x
Özcan T (2007) Characterization of Turkish Quercus L. taxa based on fatty acid compositions of the acorns. J Am Oil Chem Soc 84:653–662. https://doi.org/10.1007/s11746-007-1087-8
Paradies G, Paradies V, Ruggiero FM, Petrosillo G (2019) Role of cardiolipin in mitochondrial function and dynamics in health and disease: molecular and pharmacological aspects. Cells 8:728. https://doi.org/10.3390/cells8070728
Powell JR, Rillig MC (2018) Biodiversity of arbuscular mycorrhizal fungi and ecosystem function. New Phytol 220:1059–1075. https://doi.org/10.1111/nph.15119
Püschel D, Janoušková M, Hujslová M, Slavíková R, Gryndlerová H, Jansa J (2016) Plant–fungus competition for nitrogen erases mycorrhizal growth benefits of Andropogon gerardii under limited nitrogen supply. Ecol Evol 6:4332–4346. https://doi.org/10.1002/ece3.2207
Redecker D (2000) Specific PCR primers to identify arbuscular mycorrhizal fungi within colonized roots. Mycorrhiza 10:73–80. https://doi.org/10.1007/s005720000061
Rezanka T (1993) Polyunsaturated and unusual fatty acids from slime moulds. Phytochemistry 33:1441–1444. https://doi.org/10.1016/0031-9422(93)85106-2
Rezanka T, Kolouchova I, Sigler K (2016) Lipidomic analysis of psychrophilic yeasts cultivated at different temperatures. Biochim Biophys Acta - Mol Cell Biol Lipids 1861:1634–1642. https://doi.org/10.1016/j.bbalip.2016.07.005
Rezanka T, Rezanka M, Mezricky D, Vítová M (2020) Lipidomic analysis of diatoms cultivated with silica nanoparticles. Phytochemistry 177:112452. https://doi.org/10.1016/j.phytochem.2020.112452
Rezanka T, Lukavský J, Rozmoš M, Nedbalová L, Jansa J (2022) Separation of triacylglycerols containing positional isomers of hexadecenoic acids by enantiomeric liquid chromatography-mass spectrometry. J Chrom B 1208:123401. https://doi.org/10.1016/j.jchromb.2022.123401
Rimington WR, Pressel S, Duckett JG, Field KJ, Bidartondo MI (2019) Evolution and networks in ancient and widespread symbioses between Mucoromycotina and liverworts. Mycorrhiza 29:551–565. https://doi.org/10.1007/s00572-019-00918-x
Schlame M, Brody S, Hostetler KY (1993) Mitochondrial cardiolipin in diverse eukaryotes. Eur J Biochem 212:727–733. https://doi.org/10.1111/j.1432-1033.1993.tb17711.x
Schlame M, Rua D, Greenberg ML (2000) The biosynthesis and functional role of cardiolipin. Prog Lipid Res 39:257–288. https://doi.org/10.1016/S0163-7827(00)00005-9
Schmitz O, Danneberg G, Hundeshagen B, Klingner A, Bothe H (1991) Quantification of vesicular-arbuscular mycorrhiza by biochemical parameters. J Plant Physiol 139:106–114. https://doi.org/10.1016/S0176-1617(11)80174-4
Schwarzott D, Schüβler A (2001) A simple and reliable method for SSU rRNA gene DNA extraction, amplification, and cloning from single AM fungal spores. Mycorrhiza 10:203–207. https://doi.org/10.1007/PL00009996
Sohlenkamp C (2021) Crossing the lipid divide. J Biol Chem 297:100859. https://doi.org/10.1016/j.jbc.2021.100859
Sylvia D (1992) Quantification of external hyphae of vesicular-arbuscular mycorrhizal fungi. Meth Microbiol 24:53–65. https://doi.org/10.1016/S0580-9517(08)70086-2
Thonar C, Erb A, Jansa J (2012) Real-time PCR to quantify composition of arbuscular mycorrhizal fungal communities—marker design, verification, calibration and field validation. Mol Ecol Res 12:219–232. https://doi.org/10.1111/j.1755-0998.2011.03086.x
van der Heijden MGA, Bardgett RD, van Straalen NM (2008) The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol Lett 11:296–310. https://doi.org/10.1111/j.1461-0248.2007.01139.x
van Groenigen K-J, Bloem J, Bååth E, Boeckx P, Rousk J, Bodé S, Forristal D, Jones MB (2010) Abundance, production and stabilization of microbial biomass under conventional and reduced tillage. Soil Biol Biochem 42:48–55. https://doi.org/10.1016/j.soilbio.2009.09.023
Vickery JR (1971) The fatty acid composition of the seed oils of proteaceae: a chemotaxonomic study. Phytochemistry 10:123–130. https://doi.org/10.1016/S0031-9422(00)90259-0
Vickery J, Whitfield F, Ford G, Kennett B (1984) The fatty-acid composition of gymnospermae seed and leaf oils. J Am Oil Chem Soc 61:573–575. https://doi.org/10.1007/BF02677035
Vierheilig H, Schweiger P, Brundrett M (2005) An overview of methods for the detection and observation of arbuscular mycorrhizal fungi in roots. Phys Plant 125:393–404. https://doi.org/10.1111/j.1399-3054.2005.00564.x
Voříšková A, Jansa J, Püschel D, Krüger M, Cajthaml T, Vosátka M, Janoušková M (2017) Real-time PCR quantification of arbuscular mycorrhizal fungi: does the use of nuclear or mitochondrial markers make a difference? Mycorrhiza 27:577–585. https://doi.org/10.1007/s00572-017-0777-9
Walker R (1969) Cis-11-hexadecenoic acid from Cytophaga hutchinsonii lipids. Lipids 4:15–18. https://doi.org/10.1007/BF02531788
Walker C, Gollotte A, Redecker D (2018) A new genus, Planticonsortium (Mucoromycotina), and new combination (P. tenue), for the fine root endophyte, Glomus tenue (basionym Rhizophagus tenuis). Mycorrhiza 28:213–219. https://doi.org/10.1007/s00572-017-0815-7
Welc M, Bünemann EK, Fließbach A, Frossard E, Jansa J (2012) Soil bacterial and fungal communities along a soil chronosequence assessed by fatty acid profiling. Soil Biol Biochem 49:184–192. https://doi.org/10.1016/j.soilbio.2012.01.032
Wewer V, Brands M, Dörmann P (2014) Fatty acid synthesis and lipid metabolism in the obligate biotrophic fungus Rhizophagus irregularis during mycorrhization of Lotus japonicus. Plant J 79:398–412. https://doi.org/10.1111/tpj.12566
Wolff R, Christie W, Aitzetmuller K, Pasquier E, Pedrono F, Destaillats F, Marpeau A (2000) Arachidonic and eicosapentaenoic acids in Araucariaceae, a unique feature among seed plants. Oléagineux, Corps Gras, Lipides 7:113–117. https://doi.org/10.1051/ocl.2000.0113
Zhou Y, Peisker H, Dörmann P (2016) Molecular species composition of plant cardiolipin determined by liquid chromatography mass spectrometry. J Lipid Res 57:1308–1321. https://doi.org/10.1194/jlr.D068429
Funding
This study was supported by Institutional Research Concept (Institute of Microbiology, Prague, Czech Republic), grant number RVO61388971, Ministry of Agriculture of the Czech Republic (institutional support No. MZE-RO1923), and by the Czech Science Foundation (GAČR) project 21-07247S.
Author information
Authors and Affiliations
Contributions
TŘ conceived the study and conducted the biochemical analyses. Together with JJ, he also wrote the first draft of the manuscript. HH and JJ provided biological materials and contributed to writing. LK contributed to data analysis and writing. All authors approved the final version of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare that all relevant sources of funding have been acknowledged and that they are not aware of any circumstances that could be construed as constituting a financial or nonfinancial conflict of interest. Generative artificial intelligence tools have not been used during preparation of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Řezanka, T., Hršelová, H., Kyselová, L. et al. Can cardiolipins be used as a biomarker for arbuscular mycorrhizal fungi?. Mycorrhiza 33, 399–408 (2023). https://doi.org/10.1007/s00572-023-01129-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-023-01129-1