Abstract
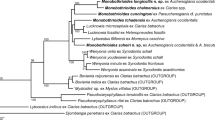
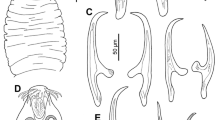
Tapeworms (Cestoda: Proteocephalidae) are the dominant component of communities of intestinal parasites in pimelodid and other catfishes (Siluriformes) from South America. Even though these parasites have been studied intensively over more than one century, molecular taxonomy and phylogenetics have questioned their morphology-based classification, thus raising doubts about the systematic value of traits commonly used to circumscribe individual taxa. In the present study, members of three morphologically well-characterized genera of proteocephalids from pimelodid (Hemisorubim platyrhynchos and Sorubim lima) and auchenipterid (Ageneiosus inermis) catfishes from the Paraná or Amazon River basins were subjected to DNA sequencing of the large subunit nuclear ribosomal RNA (lsrDNA) and complete mitochondrial cytochrome c oxidase subunit I (COI). Phylogenetic analyses revealed the sister relationship between Manaosia bracodemoca and Mariauxiella piscatorum, and among Mariauxiella pimelodi and Ageneiella brevifilis. As a result, Mar. piscatorum and A. brevifilis are transferred to Manaosia and Mariauxiella, respectively, as Manaosia piscatorum n. comb. and Mariauxiella brevifilis n. comb., and the genus Ageneiella is suppressed. Diagnoses of Manaosia and Mariauxiella are amended. In addition, the present study revealed misidentification of tapeworms whose sequences are deposited in the GenBank database.



Similar content being viewed by others
References
Alves PV, de Chambrier A, Scholz T, Luque JL (2017) Annotated checklist of fish cestodes from South America. Zookeys 650:1–205. https://doi.org/10.3897/zookeys.650.10982
Alves PV, Kuchta R, Scholz T (2019) Molecular data support monophyly of the recently erected Riggenbachiella (Cestoda: Proteocephalidae), parasites of Neotropical catfishes. Zootaxa 4706:594–597. https://doi.org/10.11646/zootaxa.4706.4.9
Alves PV, de Chambrier A, Luque JL, Scholz T (2021) Integrative taxonomy reveals hidden cestode diversity in Pimelodus catfishes in the Neotropics. Zool Scr 50:210–224. https://doi.org/10.1111/zsc.12465
Arredondo NJ, Gil de Pertierra AA (2008) The taxonomic status of Spatulifer cf. maringaensis Pavanelli & Rego, 1989 (Eucestoda: Proteocephalidea) from Sorubim lima (Bloch & Schneider) (Pisces: Siluriformes), and the use of the microthrix pattern in the discrimination of Spatulifer spp. Syst Parasitol 70:223–236. https://doi.org/10.1007/s11230-008-9142-x
Barčák D, Oros M, Hanzelová V, Scholz T (2014) Phenotypic plasticity in Caryophyllaeus brachycollis Janiszewska, 1953 (Cestoda: Caryophyllidea): does fish host play a role? Syst Parasitol 88:153–166. https://doi.org/10.1007/s11230-014-9495-2
de Chambrier A (2003) Systematic status of Manaosia bracodemoca Woodland, 1935 and Paramonticellia itaipuensis Pavanelli et Rego, 1991 (Eucestoda: Proteocephalidea), parasites of Sorubim lima (Siluriformes: Pimelodidae) from South America. Folia Parasitol 50:121–127. https://doi.org/10.14411/fp.2003.021
de Chambrier A, Rego AA (1995) Mariauxiella pimelodi n. g., n. sp. (Cestoda: Monticelliidae): a parasite of pimelodid siluroid fishes from South America. Syst Parasitol 30:57–65. https://doi.org/10.1007/BF00009245
de Chambrier A, Vaucher C (1999) Proteocephalidae et Monticellidae (Eucestoda: Proteocephalidea) parasites de poissons d’eau douce au Paraguay, avec descriptions d’un genre nouveau et de dix espèces nouvelles. Rev Suisse Zool 106:165–240. https://doi.org/10.5962/bhl.part.80074
de Chambrier A, Zehnder MP, Vaucher C, Mariaux J (2004) The evolution of the Proteocephalidea (Platyhelminthes, Eucestoda) based on an enlarged molecular phylogeny, with comments on their uterine development. Syst Parasitol 57:159–171. https://doi.org/10.1023/B:SYPA.0000019083.26876.34
de Chambrier A, Scholz T, Kuchta R, Posel P, Mortenthaler M, Guardia CC (2006) Tapeworms (Cestoda: Proteocephalidea) of fishes from the Amazon River in Peru. Comp Parasitol 73:111–120. https://doi.org/10.1654/4182.1
de Chambrier A, Waeschenbach A, Fisseha M, Scholz T, Mariaux J (2015) A large 28S rDNA-based phylogeny confirms the limitations of established morphological characters for classification of proteocephalidean tapeworms (Platyhelminthes, Cestoda). Zookeys 500:25–59. https://doi.org/10.3897/zookeys.500.9360
de Chambrier A, Scholz T, Mariaux J, Kuchta R (2017) Onchoproteocephalidea I. Caira, Jensen, Waeschenbach, Olson & Littlewood, 2014. In: Caira JN, Jensen K (eds) Planetary biodiversity inventory (2008–2017): tapeworms from vertebrate bowels of the earth. Special Publication No. 25. University of Kansas, Natural History Museum, Lawrence, KS, USA, pp 251–277
de Pertierra G (2009) Luciaella ivanovae n. g., n. sp. (Eucestoda: Proteocephalidea: Peltidocotylinae), a parasite of Ageneiosus inermis (L.) (Siluriformes: Auchenipteridae) in Argentina. Syst Parasitol 73:71–80. https://doi.org/10.1007/s11230-009-9174-x
Dereeper A, Guignon V, Blanc G, Audic S, Buffet S, Chevenet F, Dufayard JF, Guido S, Lefort V, Lescot M, Claverie JM, Gascuel O (2008) Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res 36:W465–W469. https://doi.org/10.1093/nar/gkn180
Diesing KM (1850) Systema Helminthum, vol I. Braumüller, Vienna
Freze VI (1965) [Proteocephalata in fish, amphibians and reptiles]. Essentials of Cestodology. Vol. V. Nauka, Moscow, 538 pp. (In Russian: English translation, Israel Program of Scientific Translation, 1969, Cat. No. 1853. v + 597 pp.)
Guindon S, Dufayard J-F, Lefort V, Anisimova M, Hordijk W, Gascuel O (2010) New algorithms and methods to estimate maximum likelihood phylogenies: assessing the performance of PhyML 3.0. Syst Biol 59:307–321. https://doi.org/10.1093/sysbio/syq010
Hoang DT, Chernomor O, von Haeseler A, Minh BQ, Vinh LS (2018) UFBoot2: improving the ultrafast bootstrap approximation. Mol Biol Evol 35:518–522. https://doi.org/10.1093/molbev/msx281
Jensen K, Caira JN, Cielocha JJ, Littlewood DTJ, Waeschenbach A (2016) When proglottids and scoleces conflict: phylogenetic relationships and a family-level classification of the Lecanicephalidea (Platyhelminthes: Cestoda). Int J Parasitol 46:291–310. https://doi.org/10.1016/j.ijpara.2016.02.002
Kalyaanamoorthy S, Minh BQ, TKF W, von Haeseler A, Jermiin LS (2017) ModelFinder: fast model selection for accurate phylogenetic estimates. Nat Methods 14:587–589. https://doi.org/10.1038/nmeth.4285
Katoh K, Standley DM (2013) MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol Biol Evol 30:772–780. https://doi.org/10.1093/molbev/mst010
Kearse M, Moir R, Wilson A et al (2012) Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28:1647–1649. https://doi.org/10.1093/bioinformatics/bts199
Kumar S, Stecher G, Tamura K (2016) MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874 https://doi.org/10.1093/molbev/msw054
Miller MA, Pfeiffer W, Schwartz T (2010) Creating the CIPRES science gateway for inference of large phylogenetic trees. In: Proceedings of the gateway computing environments workshop (GCE), 14 Nov. 2010, New Orleans, LA, 1–8 https://doi.org/10.1145/2016741.2016785
Minh BQ, Nguyen MAT, von Haeseler A (2013) Ultrafast approximation for phylogenetic bootstrap. Mol Biol Evol 30:1188–1195. https://doi.org/10.1093/molbev/mst024
Oros M, Uhrovič D, Choudhury A, Mackiewicz JS, Scholz T (2020) Scolex morphology of monozoic tapeworms (Caryophyllidea) from the Nearctic Region: taxonomic and evolutionary implications. J Biomed Inform 39:86–102. https://doi.org/10.1016/j.jbi.2005.08.008
Planet PJ (2006) Tree disagreement: measuring and testing incongruence in phylogenies. J Biomed Inform 39:86–102. https://doi.org/10.1016/j.jbi.2005.08.008
Pleijel F, Jondelius U, Norlinder E, Nygren A, Oxelman B, Schander C, Sundberg P, Thollesson M (2008) Phylogenies without roots? A plea for the use of vouchers in molecular phylogenetic studies. Mol Phylogenet Evol 48:369–371. https://doi.org/10.1016/j.ympev.2008.03.024
Rego AA (1994) Order Proteocephalidea Mola, 1928. In: Khalil LF, Jones A, Bray RA (eds) Keys to the cestode parasites of vertebrates. CAB International, Wallingford, pp 257–293
Rego AA, Chubb JC, Pavanelli GC (1999) Cestodes in South American freshwater teleost fishes: keys to genera and brief description of species. Rev Bras de Zool 16:299–367. https://doi.org/10.1590/S0101-8175199900020000
Ronquist F, Teslenko M, van der Mark P, Ayres DL, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. https://doi.org/10.1093/sysbio/sys029
Scholz T, de Chambrier A, Kuchta R, DTJ L, Waeschenbach A (2013) Macrobothriotaenia ficta (Cestoda: Proteocephalidea), a parasite of sunbeam snake (Xenopeltis unicolor): example of convergent evolution. Zootaxa 3640:485–499. https://doi.org/10.11646/zootaxa.3640.3.12
Sela I, Ashkenazy H, Katoh K, Pupko T (2015) GUIDANCE2: accurate detection of unreliable alignment regions accounting for the uncertainty of multiple parameters. Nucleic Acids Res 43:W7–W14. https://doi.org/10.1093/nar/gkq443
Swofford DL (2003) PAUP*: phylogenetic analysis using parsimony (and other methods). Sinauer Associates, Sunderland.
Trifinopoulos J, Nguyen LT, von Haeseler A, Minh BQ (2016) W-IQ-TREE: a faset online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res 44:W232–W235. https://doi.org/10.1093/nar/gkw256
Xia X (2018) DAMBE7: New and improved tools for data analysis in molecular biology and evolution. Mol Biol Evol 35:1550–1552. https://doi.org/10.1093/molbev/msy073
Zehnder MP, Mariaux J (1999) Molecular systematic analysis of the order Proteocephalidea (Eucestoda) based on mitochondrial and nuclear rDNA sequences. Int J Parasitol 29:1841–1852. https://doi.org/10.1016/S0020-7519(99)00122-8
Acknowledgements
The authors are indebted to Roman Kuchta (Institute of Parasitology, České Budějovice) and Carlos A. Mendoza-Palmero (Masaryk University, Brno) for collecting the specimen of M. piscatorum in Iquitos, Peru in 2009. Our thanks are extended to Jan Brabec (Institute of Parasitology, České Budějovice) for providing unpublished sequences, colleagues of the Laboratory of Fish Parasitology (UFRRJ) who helped with collection and dissection of fishes, and to Nathalia J. Arredondo (Universidad de Buenos Aires) for inspiring discussions about the interrelationships of Manaosia and Mariauxiella.
Funding
This study was supported by the “Ciência sem fronteiras” Brazilian program—visitant researcher modality (No. 135/2012) (stays of TS in Brazil at the Universidade Federal Rural de Rio de Janeiro in 2013–2015). This work was also funded by CAPES (postdoc scholarship to PVA, grant 88887.508591/2020-00), CNPq (research scholarship to JLL, grant 306952/2015-7), NSF-PBI awards (grants 0818696 and 0818823), and the Czech Science Foundation (19-28399-X).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Guillermo Salgado-Maldonado
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Alves, P.V., de Chambrier, A., Luque, J.L. et al. New arrangement of three genera of fish tapeworms (Cestoda: Proteocephalidae) in catfishes (Siluriformes) from the Neotropical Region: taxonomic implications of molecular phylogenetic analyses. Parasitol Res 120, 1593–1603 (2021). https://doi.org/10.1007/s00436-021-07138-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-021-07138-3