Abstract
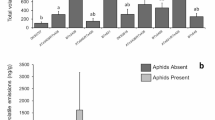
The Enemy Release Hypothesis (ERH) predicts that the escape from natural enemies, such as specialist herbivores, may explain the invasiveness of some invasive alien plants, maximizing their investment in growth and reproduction. This release from natural enemies might decrease the investment in expensive defense mechanisms (i.e., digestibility reducers) against the attack of specialist enemies, whilst increasing the investment in defenses (i.e., cheap toxins) and tolerance against the attack of generalist herbivores, as exposed by the Shifting Defense Hypothesis (SDH). To test this, we conducted a greenhouse experiment to compare morphological and physiological traits of Carpobrotus edulis, collected in its native (South Africa) and introduced (Chile and Spain) ranges, attacked by the generalist spittlebug Philaenus spumarius and the specialist scale insect Pulvinariella mesembryanthemi. Our results do not support the ER and SD hypotheses. We found that C. edulis plants collected from native and introduced ranges showed no significant differences in growth and defensive compounds studied for both controls and those plants under attack by the generalist P. spumarius (i.e., showing no increase in biomass or changes in biochemical defenses). In contrast, the specialist herbivore P. mesembryanthemi induced the production of total phenols and tannins and reduced the growth and survival of C. edulis plants. Overall, we found strong evidence that C. edulis is negatively affected by the attack of its specialist herbivore, but not by that of generalist herbivore, regardless of origin. The observed tolerance to generalist herbivores suggests the intriguing possibility of trait selection, allowing C. edulis plants to tolerate generalist herbivores more than specialists.




Similar content being viewed by others
Availability of data and materials
The datasets generated during and/or analyzed during the current study are available in the FigShare repository. https://doi.org/10.6084/m9.figshare.14604486.v1
References
Amouroux P, Crochard D, Germain J-F et al (2017) Genetic diversity of armored scales (Hemiptera: Diaspididae) and soft scales (Hemiptera: Coccidae) in Chile. Sci Rep 7:2014. https://doi.org/10.1038/s41598-017-01997-6
Ashton IW, Lerdau MT (2008) Tolerance to herbivory, and not resistance, may explain differential success of invasive, naturalized, and native North American temperate vines. Divers Distrib 14:169–178. https://doi.org/10.1111/j.1472-4642.2007.00425.x
Bartomeus I, Vilà M, Santamaría L (2008) Contrasting effects of invasive plants in plant–pollinator networks. Oecologia 155:761–770. https://doi.org/10.1007/s00442-007-0946-1
Bazzaz FA, Chiariello NR, Coley PD, Pitelka LF (1987) Allocating resources to reproduction and defense. Bioscience 37:58–67. https://doi.org/10.2307/1310178
Bewick V, Cheek L, Ball J (2004) Statistics review 12: survival analysis. Crit Care 8:389–394. https://doi.org/10.1186/cc2955
Blossey B, Notzold R (1995) Evolution of increased competitive ability in invasive nonindigenous plants: a hypothesis. J Ecol 83:887. https://doi.org/10.2307/2261425
Campoy JG, Acosta ATR, Affre L et al (2018) Monographs of invasive plants in Europe: Carpobrotus. Bot Lett 165:440–475. https://doi.org/10.1080/23818107.2018.1487884
Collins L, Scott JK (1982) Interaction of ants, predators and the scale insect, Pulvinariella mesembryanthemi, on Carpobrotus edulis, an exotic plant naturalized in Western Australia. Aust Entomol 8:73–78
Cox DR (1972) Regression models and life-tables. J Roy Stat Soc Ser B 34:187–220
D’Antonio CM, Mahall BE (1991) Root profiles and competition between the invasive, exotic perennial, Carpobrotus edulis, and two native shrub species in California coastal scrub. Am J Bot 78:885–894. https://doi.org/10.2307/2445167
Day NJ, Dunfield KE, Antunes PM (2016) Fungi from a non-native invasive plant increase its growth but have different growth effects on native plants. Biol Invasions 18:231–243. https://doi.org/10.1007/s10530-015-1004-2
de la Peña E, de Clercq N, Bonte D et al (2010) Plant-soil feedback as a mechanism of invasion by Carpobrotus edulis. Biol Invasions 12:3637–3648. https://doi.org/10.1007/s10530-010-9756-1
Doorduin LJ, Vrieling K (2011) A review of the phytochemical support for the shifting defence hypothesis. Phytochem Rev 10:99–106. https://doi.org/10.1007/s11101-010-9195-8
Earnshaw MJ, Carver KA, Charlton WA (1987) Leaf anatomy, water relations and crassulacean acid metabolism in the chlorenchyma and colourless internal water-storage tissue of Carpobrotus edulis and Senecio mandraliscae. Planta 170:421–432
Fenollosa E, Roach DA, Munné-Bosch S (2016) Death and plasticity in clones influence invasion success. Trends Plant Sci 21:551–553. https://doi.org/10.1016/j.tplants.2016.05.002
Fenollosa E, Munné-Bosch S, Pintó-Marijuan M (2017) Contrasting phenotypic plasticity in the photoprotective strategies of the invasive species Carpobrotus edulis and the coexisting native species Crithmum maritimum. Physiol Plant 160:185–200. https://doi.org/10.1111/ppl.12542
Fernandes GW (1990) Hypersensitivity: a neglected plant resistance mechanism against insect herbivores. Environ Entomol 19:1173–1182. https://doi.org/10.1093/ee/19.5.1173
Fernandes GW, Negreiros D (2001) The occurrence and effectiveness of hypersensitive reaction against galling herbivores across host taxa. Ecol Entomol 26:46–55. https://doi.org/10.1046/j.1365-2311.2001.00290.x
Folin O, Ciocalteau V (1927) Tyrosine and tryptophane in proteins. J Biol Chem 73:627–648
Franco JC, Russo A, Marotta S (2011) An annotated checklist of scale insects (Hemiptera: Coccoidea) of Portugal, including Madeira and Azores Archipelagos. Zootaxa 3004:1–32. https://doi.org/10.11646/zootaxa.3004.1.1
Fukano Y, Yahara T (2012) Changes in defense of an alien plant Ambrosia artemisiifolia before and after the invasion of a native specialist enemy Ophraella communa. PLoS ONE 7:e49114. https://doi.org/10.1371/journal.pone.0049114
Gómez-Menor Ortega J (1927) Estudio monográfico de los cóccidos de España. PhD Thesis, Universidad Complutense de Madrid, Madrid
Halkka O, Halkka L (1990) Population genetics of the polymorphic meadow spittlebug, Philaenus spumarius (L.). Evol Biol 24:149–191
Halkka O, Raatikainen M, Vasarainen A, Heinonen L (1967) Ecology and ecological genetics of Philaenus spumarius (L.) (Homoptera). Ann Bot Fenn 4:1–18
Harrington DP, Fleming TR (1982) A class of rank test procedures for censored survival data. Biometrika 69:553–566. https://doi.org/10.1093/biomet/69.3.553
Harvey KJ, Nipperess DA, Britton DR, Hughes L (2013) Does time since introduction influence enemy release of an invasive weed? Oecologia 173:493–506. https://doi.org/10.1007/s00442-013-2633-8
Herms DA, Mattson WJ (1992) The dilemma of plants: to grow or defend. Q Rev Biol 67:283–335. https://doi.org/10.1086/417659
Hierro JL, Eren Ö, Čuda J, Meyerson LA (2022) Evolution of increased competitive ability (EICA) may explain dominance of introduced species in ruderal communities. Ecol Monogr. https://doi.org/10.1002/ecm.1524
Huang W, Siemann E, Wheeler GS et al (2010) Resource allocation to defence and growth are driven by different responses to generalist and specialist herbivory in an invasive plant. J Ecol 98:1157–1167. https://doi.org/10.1111/j.1365-2745.2010.01704.x
Kaplan EL, Meier P (1958) Nonparametric estimation from incomplete samples. J Am Stat Assoc 53:457–481
Kaplan I, Halitschke R, Kessler A et al (2008) Constitutive and induced defenses to herbivory in above- and belowground plant tissues. Ecology 89:392–406. https://doi.org/10.1890/07-0471.1
Karban R, Baldwin IT (1997) Induced responses to herbivory. The University of Chicago Press, Chicago and London
Karban R, Myers JH (1989) Induced plant responses to herbivory. Annu Rev Ecol Syst 20:331–348
Keane RM, Crawley MJ (2002) Exotic plant invasions and the enemy release hypothesis. Trends Ecol Evol 17:164–170. https://doi.org/10.1016/S0169-5347(02)02499-0
Kondo T, Gullan PJ (2010) The coccidae (Hemiptera: Coccoidea) of Chile, with descriptions of three new species and transfer of Lecanium resinatum Kieffer & Herbst to the Kerriidae. Zootaxa 2560:1–15. https://doi.org/10.11646/zootaxa.2560.1.1
Kramer DM, Johnson G, Kiirats O, Edwards GE (2004) New fluorescence parameters for the determination of QA redox state and excitation energy fluxes. Photosynth Res 79:209–218. https://doi.org/10.1023/B:PRES.0000015391.99477.0d
Lenth RV (2016) Least-squares means: the r package lsmeans. J Stat Softw 69:1–33. https://doi.org/10.18637/jss.v069.i01
Lieurance D, Cipollini D (2013) Environmental influences on growth and defence responses of the invasive shrub, Lonicera maackii, to simulated and real herbivory in the juvenile stage. Ann Bot 112:741–749. https://doi.org/10.1093/aob/mct070
Lin T, Klinkhamer PGL, Vrieling K (2015) Parallel evolution in an invasive plant: Effect of herbivores on competitive ability and regrowth of Jacobaea vulgaris. Ecol Lett 18:668–676. https://doi.org/10.1111/ele.12445
Liu M, Pan X, Zhang Z et al (2004) (2020) Testing the shifting defense hypothesis for constitutive and induced resistance and tolerance. J Pest Sci 93:355–364. https://doi.org/10.1007/s10340-019-01162-0
Miller GL, Miller DR (2003) Invasive soft scales (Hemiptera: Coccidae) and their threat to U.S. agriculture. Proc Entomol Soc Wash 105:832–846
Montesinos D (2021) Fast invasives fastly become faster: invasive plants align largely with the fast side of the plant economics spectrum. J Ecol. https://doi.org/10.1111/1365-2745.13616
Müller-Schärer H, Schaffner U, Steinger T (2004) Evolution in invasive plants: implications for biological control. Trends Ecol Evol 19:417–422. https://doi.org/10.1016/j.tree.2004.05.010
Ni G, Zhao P, Ye Y et al (2020) High photosynthetic capacity and energy-use efficiency benefit both growth and chemical defense in invasive plants. Chemoecology 30:69–78. https://doi.org/10.1007/s00049-020-00299-0
Novoa A, González L, Moravcová L, Pyšek P (2012) Effects of soil characteristics, allelopathy and frugivory on establishment of the invasive plant Carpobrotus edulis and a co-ccuring native Malcolmia littorea. PLoS ONE 7:e53166. https://doi.org/10.1371/journal.pone.0053166
Novoa A, González L, Moravcová L, Pyšek P (2013) Constraints to native plant species establishment in coastal dune communities invaded by Carpobrotus edulis: implications for restoration. Biol Conserv 164:1–9. https://doi.org/10.1016/j.biocon.2013.04.008
Novoa A, Rodríguez R, Richardson DM, González L (2014) Soil quality: a key factor in understanding plant invasion? The case of Carpobrotus edulis (L.) N.E.Br. Biol Invasions 16:429–443. https://doi.org/10.1007/s10530-013-0531-y
Novoa A, Keet J-H, Lechuga-Lago Y et al (2020) Urbanization and Carpobrotus edulis invasion alter the diversity and composition of soil bacterial communities in coastal areas. FEMS Microbiol Ecol 96:finaa106. https://doi.org/10.1093/femsec/fiaa106
Núñez-González N, Rodríguez J, González L (2021) Managing the invasive plant Carpobrotus edulis: is mechanical control or specialized natural enemy more effective? J Environ Manag 298:113554. https://doi.org/10.1016/j.jenvman.2021.113554
Oduor AMO (2022) Invasive plant species that experience lower herbivory pressure may evolve lower diversities of chemical defense compounds in the exotic range. Am J Bot 109:1382–1393. https://doi.org/10.1002/ajb2.16053
Oduor AMO, Lankau RA, Strauss SY, Gómez JM (2011) Introduced Brassica nigra populations exhibit greater growth and herbivore resistance but less tolerance than native populations in the native range. New Phytol 191:536–544. https://doi.org/10.1111/j.1469-8137.2011.03685.x
Oduor AMO, van Kleunen M, Stift M (2017) In the presence of specialist root and shoot herbivory, invasive-range Brassica nigra populations have stronger competitive effects than native-range populations. J Ecol 105:1679–1686. https://doi.org/10.1111/1365-2745.12779
Pyšek P, Pergl J, Essl F et al (2017) Naturalized alien flora of the world: species diversity, taxonomic and phylogenetic patterns, geographic distribution and global hotspots of plant invasion. Preslia 89:203–274. https://doi.org/10.23855/preslia.2017.203
Quintana FJ (1956) Pulvinaria mesembryanthemi (Vallot) (Homoptera: Sternorrhyncha), nueva cochinilla para la fauna argentina y sus zooparásitos. Rev La Fac Agron Univ Nac La Plata 32:75–100
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Radville L, Chaves A, Preisser EL (2011) Variation in plant defense against invasive herbivores: evidence for a hypersensitive response in eastern hemlocks (Tsuga canadensis). J Chem Ecol 37:592–597. https://doi.org/10.1007/s10886-011-9962-z
Rapo C, Müller-Schärer H, Vrieling K, Schaffner U (2010) Is there rapid evolutionary response in introduced populations of tansy ragwort, Jacobaea vulgaris, When exposed to biological control? Evol Ecol 24:1081–1099. https://doi.org/10.1007/s10682-010-9367-4
Rodríguez J, Calbi M, Roiloa SR, González L (2018) Herbivory induced non-local responses of the clonal invader Carpobrotus edulis are not mediated by clonal integration. Sci Total Environ 633:1041–1050. https://doi.org/10.1016/j.scitotenv.2018.03.264
Rodríguez J, Thompson V, Rubido-Bará M et al (2019) Herbivore accumulation on invasive alien plants increases the distribution range of generalist herbivorous insects and supports proliferation of non-native insect pests. Biol Invasions 21:1511–1527. https://doi.org/10.1007/s10530-019-01913-1
Rodríguez J, Novoa A, Cordero-Rivera A et al (2020) Biogeographical comparison of terrestrial invertebrates and trophic feeding guilds in the native and invasive ranges of Carpobrotus edulis. NeoBiota 56:49–72. https://doi.org/10.3897/neobiota.56.49087
Rodríguez J, Cordero-Rivera A, González L (2021a) Impacts of the invasive plant Carpobrotus edulis on herbivore communities on the Iberian Peninsula. Biol Invasions 23:1425–1441. https://doi.org/10.1007/s10530-020-02449-5
Rodríguez J, Lorenzo P, González L (2021b) Phenotypic plasticity of invasive Carpobrotus edulis modulates tolerance against herbivores. Biol Invasions 23:1859–1875. https://doi.org/10.1007/s10530-021-02475-x
Rogers WE, Siemann E (2002) Effects of simulated herbivory and resource availability on native and invasive exotic tree seedlings. Basic Appl Ecol 3:297–307. https://doi.org/10.1078/1439-1791-00120
Roiloa SR, Rodríguez-Echeverría S, de la Peña E, Freitas H (2010) Physiological integration increases the survival and growth of the clonal invader Carpobrotus edulis. Biol Invasions 12:1815–1823. https://doi.org/10.1007/s10530-009-9592-3
Rojas PS (1965) Identificaciones de insectos entomófagos. Agric Técnica 25:39–40
Sakata Y, Yamasaki M, Isagi Y, Ohgushi T (2014) An exotic herbivorous insect drives the evolution of resistance in the exotic perennial herb Solidago altissima. Ecology 95:2569–2578. https://doi.org/10.1890/13-1455.1
Sotes GJ, Cavieres LA, Rodríguez R (2015) Carpobrotus edulis (L.) N.E. Br. (Aizoaceae) y su presencia en la flora de Chile. Gayana Botánica 72:149–151. https://doi.org/10.4067/S0717-66432015000100018
Strauss SY, Agrawal AA (1999) The ecology and evolution of plant tolerance to herbivory. Trends Ecol Evol 14:179–185. https://doi.org/10.1016/S0169-5347(98)01576-6
Strauss SY, Rudgers JA, Lau JA, Irwin RE (2002) Direct and ecological costs of resistance to herbivory. Trends Ecol Evol 17:278–285. https://doi.org/10.1016/S0169-5347(02)02483-7
Suehs CM, Affre L, Médail F (2004) Invasion dynamics of two alien Carpobrotus (Aizoaceae) taxa on a Mediterranean island: II. Reprod Strateg Hered (edinb) 92:550–556. https://doi.org/10.1038/sj.hdy.6800454
Therneau TM (2022) Package ‘survival’. Survival Analysis. 1–188. https://github.com/therneau/survival
Thompson V (1994) Spittlebug indicators of nitrogen-fixing plants. Ecol Entomol 19:391–398. https://doi.org/10.1111/j.1365-2311.1994.tb00257.x
Thompson V (2021) A new spittlebug of the genus Aphrophora Germar, 1821 (Hemiptera: Cercopoidea: Aphrophoridae) abundant on invasive iceplant, Carpobrotus edulis (L.) N. E. Brown (Aizoaceae), in coastal California. Pan-Pac Entomol 97:105–128
Thomson VP, Cunningham SA, Ball MC, Nicotra AB (2003) Compensation for herbivory by Cucumis sativus through increased photosynthetic capacity and efficiency. Oecologia 134:167–175. https://doi.org/10.1007/s00442-002-1102-6
Vallot JN (1829) Nouvelle Espèce de cochenilles. Comptes Rendues La Acad Des Sci Arts B-Lett Dijon 1828–1829:30–33
Vieites-Blanco C, Retuerto R, Lema M (2019) Effects of the fungus Sclerotinia sclerotiorum and the scale insect Pulvinariella mesembryanthemi on the ice plant Carpobrotus edulis from native and non-native areas: evaluation of the biocontrol potential. Biol Invasions 21:2159–2176. https://doi.org/10.1007/s10530-019-01964-4
Vieites-Blanco C, Paulo OS, Marabuto E, Lema M (2020a) Genetic variability on worldwide populations of the scale insect Pulvinariella mesembryanthemi. Biol Invasions 22:735–748. https://doi.org/10.1007/s10530-019-02125-3
Vieites-Blanco C, Retuerto R, Rodríguez Lado L, Lema M (2020b) Potential distribution and population dynamics of Pulvinariella mesembryanthemi, a promising biocontrol agent of the invasive plant species Carpobrotus edulis and C. aff. acinaciformis. Entomol Gen 40:173–185. https://doi.org/10.1127/entomologia/2020/0758
Wan J, Huang B, Yu H, Peng S (2019) Reassociation of an invasive plant with its specialist herbivore provides a test of the shifting defence hypothesis. J Ecol 107:361–371. https://doi.org/10.1111/1365-2745.13019
Wang RF, Feng YL (2016) Tolerance and resistance of invasive and native Eupatorium species to generalist herbivore insects. Acta Oecologica 77:59–66. https://doi.org/10.1016/j.actao.2016.09.001
Wang Y, Siemann E, Wheeler GS et al (2012) Genetic variation in anti-herbivore chemical defences in an invasive plant. J Ecol 100:894–904. https://doi.org/10.1111/j.1365-2745.2012.01980.x
Washburn JO, Frankie GW (1981) Dispersal of a scale insect, Pulvinariella mesembryanthemi (Homoptera: Coccoidea) on iceplant in California. Environ Entomol 10:724–727. https://doi.org/10.1093/ee/10.5.724
Washburn JO, Frankie GW (1985) Biological studies of iceplant scales, Pulvinariella mesembryanthemi and Pulvinaria delottoi (Homoptera: Coccidae), in California. Hilgardia 53:1–27
Weaver C, King D (1954) Meadow spittlebug, Philaenus leucophthalmus (L.). Ohio Agric Exp Stn Res Bull 741:1–99
Wheeler GS, Schaffner U (2013) Improved understanding of weed biological control safety and impact with chemical ecology: a review. Invasive Plant Sci Manag 6:16–29. https://doi.org/10.1614/ipsm-d-12-00032.1
Winter K (1973) NaCl-induzierter Crassulaceensäurestoffwechsel bei einer weiteren Aizoacee: Carpobrotus edulis. Planta 115:187–188. https://doi.org/10.1007/BF00387783
Wittstock U, Gershenzon J (2002) Constitutive plant toxins and their role in defense against herbivores and pathogens. Curr Opin Plant Biol 5:300–307. https://doi.org/10.1016/S1369-5266(02)00264-9
Yurtsever S (2000) On the polymorphic meadow spittlebug, Philaenus spumarius (L.) (Homoptera: Cercopidae). Turkish J Zool 24:447–459. https://doi.org/10.1080/03014223.2000.9517632
Zhang Z, Pan X, Blumenthal D et al (2018) Contrasting effects of specialist and generalist herbivores on resistance evolution in invasive plants. Ecology 99:866–875. https://doi.org/10.1002/ecy.2155
Züst T, Agrawal AA (2017) Trade-offs between plant growth and defense against insect herbivory: an emerging mechanistic synthesis. Annu Rev Plant Biol 68:513–534. https://doi.org/10.1146/annurev-arplant-042916-040856
Acknowledgements
We are grateful to Desika Moodley for their valuable comments on the manuscript and English editing. This work was carried out within the framework of the project “Retos en la gestión de la planta invasora Carpobrotus edulis. Variabilidad fenotípica y cambios en la relación suelo-planta durante el proceso de invasion” (in Spanish), Reference CGL2013-48885-C2-1-R, funded by the Ministry of Economy and Competitiveness (Spanish Government). JR acknowledges financial support from the European Union NextGenerationEU and “Plan de Recuperación, Transformación y Resiliencia” awarded by the Ministry of Universities and granted by the Universidade de Vigo (UP2021-046) through the Margarita Salas program. JR and AN acknowledge funding from the Czech Science Foundation (Project No. 19-13142S and EXPRO Grant No. 19-28807X); and the Czech Academy of Sciences (long-term research development project No. RVO 67985939). AP was funded by CONICYT PIA AFB-170008. We sincerely thank the constructive and very helpful comments provided by the two anonymous reviewers that substantially improved the final version of the manuscript.
Author information
Authors and Affiliations
Contributions
JR designed the methodology and led the writing of the manuscript; JR and GS conceived the ideas; JR, AN, GS, AP and LG collected the seeds, and JR the insects; JR performed the experiment, collected, and analyzed the data. All authors contributed to the drafts, agreed on the content and gave final approval for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rodríguez, J., Novoa, A., Sotes, G. et al. Variation in defensive traits against herbivores of native and invasive populations of Carpobrotus edulis. Biol Invasions 25, 1149–1164 (2023). https://doi.org/10.1007/s10530-022-02970-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-022-02970-9