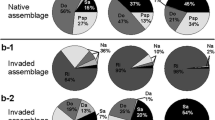
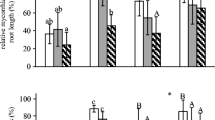
Abstract
Arbuscular mycorrhizal fungi (AMF) colonize the roots of numerous aquatic and wetland plants, but the establishment and functioning of mycorrhizal symbiosis in submerged habitats have received only little attention. Three pot experiments were conducted to study the interaction of isoetid plants with native AMF. In the first experiment, arbuscular mycorrhizal (AM) symbiosis did not establish in roots of Isoëtes echinospora and I. lacustris, while Littorella uniflora roots were highly colonized. Shoot and root biomass of L. uniflora were, however, not affected by AMF inoculation, and only one of nine AMF isolates significantly increased shoot P concentration. In the second experiment, we compared colonization by three Glomus tetrastratosum isolates of different cultivation history and origin (aquatic versus terrestrial) and their effects on L. uniflora growth and phosphorus nutrition under submerged versus terrestrial conditions. The submerged cultivation considerably slowed, but did not inhibit mycorrhizal root colonization, regardless of isolate identity. Inoculation with any AMF isolate improved plant growth and P uptake under terrestrial, but not submerged conditions. In the final experiment, we compared the communities of AMF established in two cultivation regimes of trap cultures with lake sediments, either submerged on L. uniflora or terrestrial on Zea mays. After 2-year cultivation, we did not detect a significant effect of cultivation regime on AMF community composition. In summary, although submerged conditions do not preclude the development of functional AM symbiosis, the contribution of these symbiotic fungi to the fitness of their hosts seems to be considerably less than under terrestrial conditions.





Similar content being viewed by others
Data availability
Data not included in the manuscript are accessible as a supplementary material, sequence data, and associated metadata are available from the PlutoF repository (https://plutof.ut.ee/#/doi/10.15156/BIO/807450).
References
Andersen FO, Andersen T (2006) Effects of arbuscular mycorrhizae on biomass and nutrients in the aquatic plant Littorella uniflora. Freshwater Biol 51:1623–1633
Baar J, Paradi I, Lucassen E, Hudson-Edwards KA, Redecker D, Roelofs JGM, Smolders AJP (2011) Molecular analysis of AMF diversity in aquatic macrophytes: a comparison of oligotrophic and ultra-oligotrophic lakes. Aquat Bot 94:53–61
Baldrian P, Kolařík M, Štrusová M, Kopecký J, Valášková V, Větrovský T, Žifčáková L, Šnajdr J, Ridl J, Vlček Č, Voříšková J (2012) Active and total microbial communities in forest soil are largely different and highly stratified during decomposition. ISME J 6:248–258
Ban YH, Jiang YH, Li M, Zhang XL, Zhang SY, Wu Y, Xu ZY (2017) Homogenous stands of a wetland grass living in heavy metal polluted wetlands harbor diverse consortia of arbuscular mycorrhizal fungi. Chemosphere 181:699–709
Bao XZ, Wang YT, Olsson PA (2019) Arbuscular mycorrhiza under water—carbon-phosphorus exchange between rice and arbuscular mycorrhizal fungi under different flooding regimes. Soil Biol Biochem 129:169–177
Beck-Nielsen D, Madsen TV (2001) Occurrence of vesicular-arbuscular mycorrhiza in aquatic macrophytes from lakes and streams. Aquat Bot 71:141–148
Błaszkowski J, Chwat G, Goralska A, Bobrowska-Chwat A (2015) Glomus tetrastratosum, a new species of arbuscular mycorrhizal fungi (Glomeromycota). Mycoscience 56:280–286
Borcard D, Legendre P (2002) All-scale spatial analysis of ecological data by means of principal coordinates of neighbour matrices. Ecol Model 153:51–68
Boston HL, Adams MS (1987) Productivity, growth and photosynthesis of two small ʻisoetidʼ plants, Litorella uniflora and Isoetes macrospora. J Ecol 75:333–350
Carvalho LM, Correia PM, Cacador I, Martins-Loucao MA (2003) Effects of salinity and flooding on the infectivity of salt marsh arbuscular mycorrhizal fungi in Aster tripolium L. Biol Fert Soils 38:137–143
Chen XW, Wu FY, Li H, Chan WF, Wu SC, Wong MH (2017) Mycorrhizal colonization status of lowland rice (Oryza sativa L.) in the southeastern region of China. Environ Sci Pollut Res 6:5268–5276
Christensen KK, Jensen HS, Andersen FO, Wigand C, Holmer M (1998) Interferences between root plaque formation and phosphorus availability for isoetids in sediments of oligotrophic lakes. Biogeochemistry 43:107–128
De Caceres M, Legendre P (2009) Associations between species and groups of sites: indices and statistical inference. Ecology 90:3566–3574
Deepika S, Kothamasi D (2015) Soil moisture-a regulator of arbuscular mycorrhizal fungal community assembly and symbiotic phosphorus uptake. Mycorrhiza 25:67–75
Doubková P, Suda J, Sudová R (2012) The symbiosis with arbuscular mycorrhizal fungi contributes to plant tolerance to serpentine edaphic stress. Soil Biol Biochem 44:56–64
Dunham RM, Ray AM, Inouye RS (2003) Growth, physiology, and chemistry of mycorrhizal and nonmycorrhizal Typha latifolia seedlings. Wetlands 23:890–896
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996–998
Farmer AM (1985) The occurrence of vesicular-arbuscular mycorrhiza in isoetid type submerged aquatic macrohytes under naturally varying conditions. Aquat Bot 21:245–249
Froslev TG, Kjoller R, Bruun HH, Ejrnaes R, Brunbjerg AK, Pietroni C, Hansen AJ (2017) Algorithm for post-clustering curation of DNA amplicon data yields reliable biodiversity estimates. Nat Commun 8:11
Fusconi A, Mucciarelli M (2018) How important is arbuscular mycorrhizal colonization in wetland and aquatic habitats? Env Exp Bot 155:128–141
Garcia I, Mendoza R, Pomar MC (2008) Deficit and excess of soil water impact on plant growth of Lotus tenuis by affecting nutrient uptake and arbuscular mycorrhizal symbiosis. Plant Soil 304:117–131
Goslee SC, Urban DL (2007) The ecodist package for dissimilarity-based analysis of ecological data. J Stat Softw 22:1–19
Ipsilantis I, Sylvia DM (2007) Interactions of assemblages of mycorrhizal fungi with two Florida wetland plants. Appl Soil Ecol 35:261–271
Jayachandran K, Shetty KG (2003) Growth response and phosphorus uptake by arbuscular mycorrhizae of wet prairie sawgrass. Aquat Bot 76:281–290
Katoh K, Asimenos G, Toh H (2009) Multiple alignment of DNA sequences with MAFFT. In: Posada D (ed) Bioinformatics for DNA sequence analysis, vol 537. Humana Press, Methods in Molecular Biology, pp 39–64
Klavsen SK, Madsen TV, Maberly SC (2011) Crassulacean acid metabolism in the context of other carbon-concentrating mechanisms in freshwater plants: a review. Photosynth Res 109:269–279
Kohout P, Sudová R, Janoušková M, Čtvrtlíková M, Hejda M, Pánková H, Slaviková R, Štajerová K, Vosátka M, Sýkorová Z (2014) Comparison of commonly used primer sets for evaluating arbuscular mycorrhizal fungal communities: is there a universal solution? Soil Biol Biochem 68:482–493
Koljalg U, Nilsson RH, Abarenkov K, Tedersoo L, Taylor AFS, Bahram M, Bates ST, Bruns TD, Bengtsson-Palme J, Callaghan TM et al (2013) Towards a unified paradigm for sequence-based identification of fungi. Mol Ecol 22:5271–5277
Koske RE, Gemma JN (1989) A modified procedure for staining roots to detect VA mycorrhizas. Mycol Res 92:486–505
Krüger M, Krüger C, Walker C, Stockinger H, Schüssler A (2012) Phylogenetic reference data for systematics and phylotaxonomy of arbuscular mycorrhizal fungi from phylum to species level. New Phytol 193:970–984
Krüger M, Stockinger H, Krüger C, Schüssler A (2009) DNA-based species level detection of Glomeromycota: one PCR primer set for all arbuscular mycorrhizal fungi. New Phytol 183:212–223
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129:271–280
Loreti E, van Veen H, Perata P (2016) Plant responses to flooding stress. Curr Opin Plant Biol 33:64–74
Lumini E, Vallino M, Alguacil MM, Romani M, Bianciotto V (2011) Different farming and water regimes in Italian rice fields affect arbuscular mycorrhizal fungal soil communities. Ecol Appl 21:1696–1707
Mader P, Száková J, Miholová D (1998) Classical dry ashing of biological and agricultural materials. Part II. Losses of analytes due to their retention in an insoluble residue. Analusis 26:121–129
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular arbuscular mycorrhizal fungi. New Phytol 115:495–501
Mehlich A (1984) Mehlich-3 soil test extractant: a modification of Mehlich-2 extractant. Commun Soil Sci Plan 15:1409–1416
Mendoza R, Escudero V, Garcia I (2005) Plant growth, nutrient acquisition and mycorrhizal symbioses of a waterlogging tolerant legume (Lotus glaber Mill.) in a saline-sodic soil. Plant Soil 275:305–315
Miller SP (2000) Arbuscular mycorrhizal colonization of semi-aquatic grasses along a wide hydrologic gradient. New Phytol 145:145–155
Miller SP, Bever JD (1999) Distribution of arbuscular mycorrhizal fungi in stands of the wetland grass Panicum hemitomon along a wide hydrologic gradient. Oecologia 119:586–592
Møller CL, Kjøller R, Sand-Jensen K (2013) Organic enrichment of sediments reduces arbuscular mycorrhizal fungi in oligotrophic lake plants. Freshwater Biol 58:769–779
Moora M, Opik M, Davison J, Jairus T, Vasar M, Zobel M, Eckstein RL (2016) AM fungal communities inhabiting the roots of submerged aquatic plant Lobelia dortmanna are diverse and include a high proportion of novel taxa. Mycorrhiza 26:735–745
Ocampo JA, Martin J, Hayman DS (1980) Influence of plant interactions on vesicular-arbuscular mycorrhizal infections. I. Host and non-host plants grown together. New Phytol 84:27–35
Ohsowski BM, Zaitsoff PD, Öpik M, Hart MM (2014) Where the wild things are: looking for uncultured Glomeromycota. New Phytol 204:171–179
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O’Hara RB et al (2018) (2018) Vegan: Community Ecology Package. R Package Version 2(5–3):4–6
Nielsen KB, Kjoller R, Olsson PA, Schweiger PF, Andersen FO, Rosendahl S (2004) Colonisation and molecular diversity of arbuscular mycorrhizal fungi in the aquatic plants Littorella uniflora and Lobelia dortmanna in southern Sweden. Mycol Res 108:616–625
Nielsen SL, Sand-Jensen K (1997) Growth rates and morphological adaptations of aquatic and terrestrial forms of amphibious Littorella uniflora (L.) Aschers. Plant Ecol 129:135–140
Nilsson RH, Abarenkov K, Veldre V, Nylinder S, De Wit P, Brosche S, Alfredsson JF, Ryberg M, Kristiansson E (2010) An open source chimera checker for the fungal ITS region. Mol Ecol Resour 10:1076–1081
Pinheiro J, Bates D, DebRoy S, Sarkar D and R Core Team (2018) nlme: linear and nonlinear mixed effects models. R package version 3.1–137
Ramírez-Viga TK, Aguilar R, Catillo-Arguero S, Chiappa-Carrara X, Guadarrama P, Ramos-Zapata J (2018) Wetland plant species improve performance when inoculated with arbuscular mycorrhizal fungi: a meta-analysis of experimental pot studies. Mycorrhiza 28:477–493
Sauter M (2013) Root responses to flooding. Curr Opin Plant Biol 16:282–286
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ et al (2009) Introducing mothur: Open-Source, Platform-Independent, Community-Supported Software for Describing and Comparing Microbial Communities. Appl Environ Microb 75:7537–7541
Smith SE, Gianinazzi- Pearson V (1990) Phosphate uptake and arbuscular activity in mycorrhizal Allium cepa L.: effects of photon irradiance and phosphate nutrition. Aust J Plant Physiol 17:177–188
Smolders AJP, Lucassen ECHET, Roelofs JGM (2002) The isoetid environment: biogeochemistry and threats. Aquat Bot 73:325–350
Solaiman MZ, Hirata H (1997) Effect of arbuscular mycorrhizal fungi inoculation of rice seedlings at the nursery stage upon performance in the paddy field and greenhouse. Plant Soil 191:1–12
Stevens KJ, Wall CB, Janssen JA (2011) Effects of arbuscular mycorrhizal fungi on seedling growth and development of two wetland plants, Bidens frondosa L., and Eclipta prostrata (L.) L., grown under three levels of water availability. Mycorrhiza 21:279–288
Sudová R, Kohout P, Rydlová J, Čtvrtlíková M, Suda J, Voříšková J, Kolaříková Z (2020) Diverse fungal communities associated with the roots of isoetid plants are structured by host plant identity. Fungal Ecol 45:100914
Sudová R, Rydlová J, Čtvrtlíková M, Havránek P, Adamec L (2011) The incidence of arbuscular mycorrhiza in two submerged Isoetes species. Aquat Bot 94:183–187
Sýkorová Z, Ineichen K, Wiemken A, Redecker D (2007) The cultivation bias: different communities of arbuscular mycorrhizal fungi detected in roots from the field, from bait plants transplanted to the field, and from a greenhouse trap experiment. Mycorrhiza 18:1–14
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: Molecular Evolutionary Genetics Analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Tanner CC, Clayton JS (1985) Effects of vesicular-arbuscular mycorrhizas on growth and nutrition of a submerged aquatic plant. Aquat Bot 22:377–386
Tedersoo L, Nilsson RH, Abarenkov K, Jairus T, Sadam A, Saar I, Bahram M, Bechem E, Chuyong G, Koljalg U (2010) 454 Pyrosequencing and Sanger sequencing of tropical mycorrhizal fungi provide similar results but reveal substantial methodological biases. New Phytol 188:291–301
Tisserant B, Gianinazzi-Pearson V, Gianinazzi S, Gollotte A (1993) In planta histochemical staining of fungal alkaline phosphatase activity for analysis of efficient arbuscular mycorrhizal infoections. Mycol Res 97:245–250
Vallino M, Fiorilli V, Bonfante P (2014) Rice flooding negatively impacts root branching and arbuscular mycorrhizal colonization, but not fungal viability. Plant Cell Environ 37:557–572
Větrovský T, Baldrian P, Morais D (2018) SEED 2: a user-friendly platform for amplicon high-throughput sequencing data analyses. Bioinformatics 34:2292–2294
Wang YT, Huang YL, Qiu Q, Xin GR, Yang ZY, Shi SH (2011) Flooding greatly affects the diversity of arbuscular mycorrhizal fungi communities in the roots of wetland plants. PLoS ONE 6(9):e24512
Wang YT, Li YW, Bao XZ, Bjorn LO, Li SS, Olsson PA (2016) Response differences of arbuscular mycorrhizal fungi communities in the roots of an aquatic and a semiaquatic species to various flooding regimes. Plant Soil 403:361–373
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR Protocols: A Guide to Methods and Applications. Academic Press Inc, New York, pp 315–322
Wigand C, Andersen FØ, Christensen KK, Holmer M, Jensen HS (1998) Endomycorrhizae of isoetids along a biogeochemical gradient. Limnol Oceanogr 43:508–515
Wirsel SGR (2004) Homogenous stands of a wetland grass harbour diverse consortia of arbuscular mycorrhizal fungi. FEMS Microbiol Ecol 48:129–138
Wolfe BE, Weishampel PA, Klironomos JN (2006) Arbuscular mycorrhizal fungi and water table affect wetland plant community composition. J Ecol 94:905–914
Xu ZY, Wu Y, Xiao Z, Ban YH, Belvett N (2019) Positive effects of Funneliformis mosseae inoculation on reed seedlings under water and TiO2 nanoparticles stresses. World J Microb Biot 35:81
Acknowledgments
We greatly appreciate the statistical advice of Zuzana Münzbergová and technical assistance of Miroslava Ševcová (Institute of Botany, Czech Academy of Sciences). We are also indebted to the teams of Analytical Laboratories of the Institute of Botany of the Czech Academy of Sciences for chemical analyses of plant biomass and substrates. We thank David Janos (editor) and the two anonymous reviewers for their helpful and constructive comments on a previous version of the manuscript.
Funding
This work was supported by the Czech Science Foundation (project P504/10/0781 to RS) and by the long-term research project of the Czech Academy of Sciences, Institute of Botany (RVO 67985939).
Author information
Authors and Affiliations
Contributions
The study was designed by RS. MČ collected lake sediments and cultivated isoetid plantlets. RS and JR isolated aquatic AMF strains that were then identified by FO. The experiments were performed by RS, JR, MČ, and ZK. RS, ZK, and PK analysed the data; JV performed 454-sequencing. The paper was written by RS and ZK, with contributions from all co-authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sudová, R., Rydlová, J., Čtvrtlíková, M. et al. Symbiosis of isoetid plant species with arbuscular mycorrhizal fungi under aquatic versus terrestrial conditions. Mycorrhiza 31, 273–288 (2021). https://doi.org/10.1007/s00572-020-01017-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-020-01017-y