Abstract
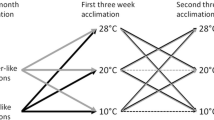
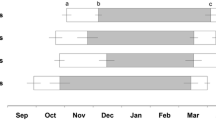
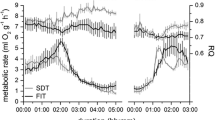
The genus Phodopus consists of three species—P. campbelli (Pc), P. sungorus (Ps), and P. roborovskii (Pr). They inhabit steppes, semi-deserts, and deserts in continental Asia with a climate changing from a moderate to a hard Continental one with extreme daily and seasonal variations. These different environmental challenges are likely to have consequences for hamsters’ morphology, physiology, and behavior. Hamsters of all three species were investigated during the course of the year in the laboratory though using natural lighting and temperature conditions. Motor activity and body temperature were measured continuously, and body mass, testes size, and fur coloration every 1–2 weeks. With regard to the pattern of activity, nearly twice as many Pc as Ps hamsters (25 vs. 14 %) failed to respond to changes of photoperiod, whereas all Pr hamsters did. Body mass and testes size were high in summer and low in winter, with the biggest relative change in Ps and the lowest in Pr hamsters. Changes of fur coloration were found in Ps hamsters only. All responding animals (that is excluding Pr), exhibited regular torpor bouts during the short winter days. In autumn, seasonal changes started considerably earlier in Ps hamsters. To investigate the putative causes of these different time courses, a further experiment was performed, to identify the critical photoperiod. Hamsters were kept for 10 weeks under different photoperiods, changing from 16 to 8 h light per day. Motor activity was recorded continuously, to identify responding and non-responding animals. Body mass was measured at the beginning and the end of the experiment, testes mass only at the end. The critical photoperiod was found to be similar in all three species. Though in a further experiment, Pc and Pr hamsters showed a delayed response, whereas the changes in Ps hamsters started immediately following transfer to short-day conditions. The results show that interspecific differences in seasonal adaptation exist, even between the closely related Ps and Pc hamsters, possibly due to different conditions in their natural habitat. Also, the impact of environmental factors like climatic conditions and food resources may differ between species.









Similar content being viewed by others
References
Bernard DJ, Losee-Olson S, Turek FW (1997) Age-related changes in the photoperiodic response of Siberian hamsters. Biol Reprod 57(1):172–177
Carter DS, Goldman BD (1983) Antigonadal effects of timed melatonin infusion in pinealectomized male Djungarian hamsters (Phodopus sungorus sungorus): duration is the critical parameter. Endocrinology 113(4):1261–1267
Ebling FJ (1994) Photoperiodic differences during development in the dwarf hamsters Phodopus sungorus and Phodopus campbelli. Gen Comp Endocrinol 95(3):475–482
Feoktistova NY (2008) Dwarf hamsters (Phodopus:Cricetinae): systematics, phylogeography, ecology, physiology, behaviour, chemical communication. [In Russian]. KMK Scientific Press Ltd., Moscow
Feoktistova NY, Meschersky IG (2005) Seasonal changes in desert hamster Phodopus roborovskii breeding activity. Acta Zoologica Sinica 51:1–6
Figala J, Hoffmann K, Goldau G (1973) The annual cycle in the Djungarian Hamster Phodopus sungorus Pallas [German]. Oecologia 12(2):89–118
Freeman DA, Goldman BD (1997) Photoperiod nonresponsive Siberian hamsters: effect of age on the probability of nonresponsiveness. J Biol Rhythms 12(2):110–121
Gorman MR, Freeman DA, Zucker I (1997) Photoperiodism in hamsters: abrupt versus gradual changes in day length differentially entrain morning and evening circadian oscillators. J Biol Rhythms 12(2):122–135
Hahn TP, Pereyra ME, Sharbaugh SM, Bentley GE (2004) Physiological responses to photoperiod in three cardueline finch species. Gen Comp Endocrinol 137(1):99–108
Hamann U (1987) Zu Aktivität und Verhalten von drei Taxa der Zwerghamster der Gattung Phodopus Miller, 1910. Zeitschrift fur Saugetierkunde-International Journal of Mammalian Biology 52(2):65–76
Heldmaier G, Steinlechner S (1981a) Seasonal control of energy-requirements for thermoregulation in the djungarian hamster (Phodopus-Sungorus), living in natural photoperiod. J Comp Physiol B 142(4):429–437
Heldmaier G, Steinlechner S (1981b) Seasonal pattern and energetics of short daily torpor in the Djungarian hamster. Phodopus-Sungorus. Oecologia 48(2):265–270
Hoffmann K (1973) Influence of photoperiod and melatonin on testis size, body-weight, and pelage color in Djungarian hamster (phodopus-sungorus). J Comp Physiol 85(3):267–282
Hoffmann K (1978) Effects of short photoperiods on puberty, growth and moult in the Djungarian hamster (Phodopus sungorus). J Reprod Fertil 54(1):29–35
Hoffmann K (1979) Photoperiod, pineal, melatonin and reproduction in hamsters. Prog Brain Res 52:397–415
Hoffmann K (1982) The critical photoperiod in the Djungarian hamster Phodopus sungorus. In: Aschoff J, Daan S, Groos G (eds) Circadian systems: structure and function. Springer Verlag, Heidelberg New York, pp 297–304
Hoffmann K, Illnerova H (1986) Photoperiodic effects in the Djungarian hamster. Rate of testicular regression and extension of pineal melatonin pattern depend on the way of change from long to short photoperiods. Neuroendocrinology 43(3):317–321
Hoffmann K, Illnerova H, Vanecek J (1986) Change in duration of the nighttime melatonin peak may be a signal driving photoperiodic responses in the Djungarian hamster (Phodopus sungorus). Neurosci Lett 67(1):68–72
Jefimow M (2007) Effects of summer- and winter-like acclimation on the thermoregulatory behavior of fed and fasted desert hamsters. Phodopus roborovskii J Thermal Biol 32:212–219
Korf HW, Schomerus C, Stehle JH (1998) The pineal organ, its hormone melatonin, and the photoneuroendocrine system. Adv Anat Embryol Cell Biol 146:1–100
Kortner G, Geiser F (2000) The temporal organization of daily torpor and hibernation: circadian and circannual rhythms. Chronobiol Int 17(2):103–128
Larkin JE, Freeman DA, Zucker I (2001) Low ambient temperature accelerates short-day responses in Siberian hamsters by altering responsiveness to melatonin. J Biol Rhythms 16(1):76–86
Masson-Pevet M, Naimi F, Canguilhem B, Saboureau M, Bonn D, Pevet P (1994) Are the annual reproductive and body weight rhythms in the male European hamster (Cricetus cricetus) dependent upon a photoperiodically entrained circannual clock? J Pineal Res 17(4):151–163
Mercer JG, Duncan JS, Lawrence CB, Trayhurn P (1994) Effect of photoperiod on mitochondrial GDP binding and adenylate cyclase activity in brown adipose tissue of Djungarian hamsters. Physiol Behav 56(4):737–740
Meschersky IG (1993) On the Difference in Thermoregulation in 2 Species of Hamsters of the Genus Phodopus [In Russian]. Zoologichesky Zhurnal 72(10):146–151
Nelson RJ (1985) Photoperiodic regulation of reproductive development in male prairie voles: influence of laboratory breeding. Biol Reprod 33(2):418–422
Neumann K, Michaux J, Lebedev V, Yigit N, Colak E, Ivanova N, Poltoraus A, Surov A, Markov G, Maak S, Neumann S, Gattermann R (2006) Molecular phylogeny of the Cricetinae subfamily based on the mitochondrial cytochrome b and 12S rRNA genes and the nuclear vWF gene. Mol Phylogenet Evol 39(1):135–148
Paul MJ, Zucker I, Schwartz WJ (2008) Tracking the seasons: the internal calendars of vertebrates. Philos Trans R Soc Lond B Biol Sci 363(1490):341–361
Petrovski DV, Novikov EA, Moshkin MP (2008) Dynamics of body temperature in the common mole vole (Ellobius talpinus, RODENTIA, CRICETIDAE) in winter period. [In Russian]. Russ J Zool 87:1504–1508
Puchalski W, Lynch GR (1986) Evidence for differences in the circadian organization of hamsters exposed to short day photoperiod. Journal of Comparative Physiology [A] 159(1):7–11
Puchalski W, Lynch GR (1994) Photoperiodic time measurement in Djungarian hamsters evaluated from T-cycle studies. Am J Physiol 267(1 Pt 2):R191–R201
Reiter RJ (1993) The melatonin rhythm: both a clock and a calendar. Experientia 49(8):654–664
Ruby NF, Joshi N, Heller HC (2002) Constant darkness restores entrainment to phase-delayed Siberian hamsters. Am J Physiol 283(6):R1314–R1320
Ruf T, Stieglitz A, Steinlechner S, Blank JL, Heldmaier G (1993) Cold exposure and food restriction facilitate physiological responses to short photoperiod in Djungarian hamsters (Phodopus sungorus). J Exp Zool 267(2):104–112
Scherbarth F, Steinlechner S (2008) The annual activity pattern of djungarian hamsters (Phodopus sungorus) is affected by wheel-running activity. Chronobiol Int 25(6):905–922
Scherbarth F, Rozman J, Klingenspor M, Brabant G, Steinlechner S (2007) Wheel running affects seasonal acclimatization of physiological and morphological traits in the Djungarian hamster (Phodopus sungorus). Am J Physiol Regul Integr Comp Physiol 293(3):R1368–R1375
Scherbarth F, Petri I, Steinlechner S (2008) Effects of wheel running on photoperiodic responses of Djungarian hamsters (Phodopus sungorus). J Comp Physiol [B] 178(5):607–615
Schottner K, Schmidt M, Hering A, Schatz J, Weinert D (2012) Short-day response in djungarian hamsters of different circadian phenotypes. Chronobiol Int 29(4):430–442
Steinlechner S, Puchalski W (2003) Mechanisms for seasonal control of reproduction in small mammals. In: Heldmaier G, Werner D (eds) Environmental signal processing and adaptation. Springer, Berlin Heidelberg, pp 233–250
Timonin ME, Place NJ, Wanderi E, Wynne-Edwards KE (2006) Phodopus campbelli detect reduced photoperiod during development but, unlike Phodopus sungorus, retain functional reproductive physiology. Reproduction 132(4):661–670
Ushakova MV, Kropotkina MV, Feoktistova NY, Surov AV (2012) Daily Torpor in Hamsters (Rodentia, Cricetinae). Russian Journal of Ecology 43(1):62–66
Vasilieva NY, Parfenova VM (2003) Does Campbell´s hamster Phodopus campbelli hibernate in winter? Experimental approach to the problem [in Russian]. In: Teriologicheskie Issledovania., vol 3. Zoological Institute of Russian Academy of Sciences, St. Petersburg, pp 105 - 123
Weiner J, Heldmaier G (1987) Metabolism and Thermoregulation in 2 Races of Djungarian Hamsters - Phodopus-Sungorus-Sungorus and Phodopus-Sungorus-Campbelli. Comparative Biochemistry and Physiology A-Physiology 86(4):639–642
Weinert D (2008) Circadian rhythms in Phodopus. Abhandlungen der Sächsischen Akademie der Wissenschaften Mathem Naturw Klasse 64(5):77–84
Weinert D, Schöttner K (2007) An inbred lineage of Djungarian hamsters with a strongly attenuated ability to synchronize. Chronobiol Int 24(6):1065–1079
Weinert D, Uhlemann S, Eimert H (1994) Age-depending effects of narcosis and transmitter implantation on circadian body temperature and activity rhythms in mice. Biol Rhythm Res 25(2):203–211
Weinert D, Schöttner K, Surov AV, Fritzsche P, Feoktistova NY, Ushakova MV, Ryurikov GB (2009) Circadian activity rhythms of dwarf hamsters (Phodopus spp.) under laboratory and semi-natural conditions. Russian Journal of Theriology 8 (1):47-58
Wynne-Edwards KE, Surov AV, Telitzina AY (1999) Differences in endogenous activity within the genus Phodopus. J Mammal 80(3):855–865
Acknowledgments
The authors are deeply grateful to four anonymous referees for their critical comments and most useful suggestions that helped us to improve the manuscript. Many thanks also to Jim Waterhouse for critical reading of the revised text.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by G. Heldmaier.
Rights and permissions
About this article
Cite this article
Müller, D., Hauer, J., Schöttner, K. et al. Seasonal adaptation of dwarf hamsters (Genus Phodopus): differences between species and their geographic origin. J Comp Physiol B 185, 917–930 (2015). https://doi.org/10.1007/s00360-015-0926-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00360-015-0926-4