Abstract
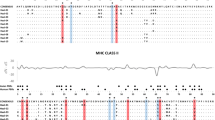
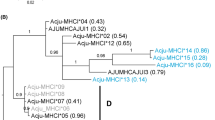
Major histocompatibility complex class II (MHC II) is an essential molecule triggering the adaptive immune response by the presentation of pathogens to helper T cells. The association between individual MHC II variants and various parasites has become a frequent finding in studies of vertebrate populations. However, although bird ectoparasites have a significant effect on their host’s fitness, and the host’s immune system can regulate ectoparasitic infections, no study has yet investigated the association between MHC II polymorphism and ectoparasite infection in the populations of free-living birds. Here, we test whether an association exists between the abundance of a chewing louse (Myrsidea nesomimi) and MHC II polymorphism of its hosts, the Galápagos mockingbirds (Mimus). We have found that the presence of two MHC II supertypes (functionally differentiated clusters) was significantly associated with louse abundance. This pattern supports the theory that a co-evolutionary interaction stands behind the maintenance of MHC polymorphism. Moreover, we have found a positive correlation between louse abundance and heterophil/lymphocyte ratio (an indicator of immunological stress) that serves as an additional piece of evidence that ectoparasite burden is affected by immunological state of Galápagos mockingbirds.

Similar content being viewed by others
Data availability
The dataset generated during the current study is available in the Mendeley Data repository, https://data.mendeley.com/datasets/75rn24pbvz/1
References
Aguilar A, Roemer G, Debenham S, Binns M, Garcelon D, Wayne RK (2004) High MHC diversity maintained by balancing selection in an otherwise genetically monomorphic mammal. Proc Natl Acad Sci U S A 101:3490–3494. https://doi.org/10.1073/pnas.0306582101
Apanius V, Penn D, Slev PR et al (1997) The nature of selection on the major histocompatibility complex. Crit Rev Immunol 17:179–224. https://doi.org/10.1615/CritRevImmunol.v17.i2.40
Bates D, Maechler M, Bolker B et al (2015) Fitting linear mixed-effects models using {lme4}. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Bateson ZW, Hammerly SC, Johnson JA, Morrow ME, Whittingham LA, Dunn PO (2016) Specific alleles at immune genes, rather than genome-wide heterozygosity, are related to immunity and survival in the critically endangered Attwater’s prairie-chicken. Mol Ecol 25:4730–4744. https://doi.org/10.1111/mec.13793
Bollmer JL, Dunn PO, Whittingham LA, Wimpee C (2010) Extensive MHC class II B gene duplication in a passerine, the common yellowthroat (Geothlypis trichas). J Hered 101:448–460. https://doi.org/10.1093/jhered/esq018
Bolnick DI, Snowberg LK, Caporaso JG et al (2014) Major histocompatibility complex class IIb polymorphism influences gut microbiota composition and diversity. Mol Ecol 23:4831–4845. https://doi.org/10.1111/mec.12846
Buczek M, Okarma H, Demiaszkiewicz AW, Radwan J (2016) MHC, parasites and antler development in red deer: no support for the Hamilton & Zuk hypothesis. J Evol Biol 29:617–632. https://doi.org/10.1111/jeb.12811
Clayton DH, Bush SE, Johnson KP (2015) Coevolution of life on hosts. University of Chicago Press, Chicago
Cohen J (1988) Statistical power analysis for the behavioral sciences. Lawrence Erlbaum Associates, New York
Cohen S, Greenwood MT, Fowler JA (1991) The louse Trinoton anserinum (Amblycera: Phthiraptera), an intermediate host of Sarconema eurycerca (Filarioidea: Nematoda), a heartworm of swans. Med Vet Entomol 5:101–110. https://doi.org/10.1111/j.1365-2915.1991.tb00527.x
Davis AK, Cook KC, Altizer S (2004) Leukocyte profiles in wild house finches with and without mycoplasmal conjunctivitis, a recently emerged bacterial disease. EcoHealth 1:362–373. https://doi.org/10.1007/s10393-004-0134-2
Davis AK, Maney DL, Maerz JC (2008) The use of leukocyte profiles to measure stress in vertebrates: a review for ecologists. Funct Ecol 22:760–772. https://doi.org/10.1111/j.1365-2435.2008.01467.x
Deem SL, Parker PG, Cruz MB et al (2011) Comparison of blood values and health status of Floreana mockingbirds (Mimus trifasciatus) on the islands of Champion and Gardner-by-Floreana, Galápagos Islands. J Wildl Dis 47:94–106. https://doi.org/10.7589/0090-3558-47.1.94
Doherty PC, Zinkernagel RM (1975) Enhanced immunological surveillance in mice heterozygous at the H-2 gene complex. Nature 256:50–52. https://doi.org/10.1038/256050a0
Doytchinova IA, Flower DR (2005) In silico identification of supertypes for class II MHCs. J Immunol 174:7085–7095. https://doi.org/10.4049/jimmunol.174.11.7085
Dunn PO, Bollmer JL, Freeman-Gallant CR, Whittingham LA (2013) MHC variation is related to a sexually selected ornament, survival, and parasite resistance in common yellowthroats. Evolution 67:679–687. https://doi.org/10.1111/j.1558-5646.2012.01799.x
Ejsmond MJ, Radwan J (2015) Red queen processes drive positive selection on major histocompatibility complex (MHC) genes. PLoS Comput Biol 11:e1004627. https://doi.org/10.1371/journal.pcbi.1004627
Fairn ER, McLellan NR, Shutler D (2012) Are lice associated with ring-billed gull chick immune responses? Waterbirds 35:164–169. https://doi.org/10.1675/063.035.0118
Haribal M, Dhondt AA, Rosane D, Rodriguez E (2005) Chemistry of preen gland secretions of passerines: different pathways to same goal? Why? Chemoecology 15:251–260. https://doi.org/10.1007/s00049-005-0318-4
Hoeck PEA, Keller LF (2012) Inbreeding, immune defence and ectoparasite load in different mockingbird populations and species in the Galápagos Islands. J Avian Biol 43:423–434. https://doi.org/10.1111/j.1600-048X.2012.05725.x
Hoeck PEA, Bucher TB, Wandeler P, Keller LF (2009) Microsatellite primers for the four Galápagos mockingbird species (Mimus parvulus, Mimus macdonaldi, Mimus melanotis and Mimus trifasciatus). Mol Ecol Resour 9:1538–1541. https://doi.org/10.1111/j.1755-0998.2009.02704.x
Hoeck PEA, Bollmer JL, Parker PG, Keller LF (2010) Differentiation with drift: a spatio-temporal genetic analysis of Galapagos mockingbird populations (Mimus spp.). Philos Trans R Soc Lond Ser B Biol Sci 365:1127–1138. https://doi.org/10.1098/rstb.2009.0311
Hoi H, Krištofík J, Darolová A, Hoi C (2012) Experimental evidence for costs due to chewing lice in the European bee-eater (Merops apiaster). Parasitology 139:53–59. https://doi.org/10.1017/S0031182011001727
Jacob J, Ziswiler W (1982) Avian biology: the uropygial gland. Academic Press, New York
Jacob S, Immer A, Leclaire S, Parthuisot N, Ducamp C, Espinasse G, Heeb P (2014) Uropygial gland size and composition varies according to experimentally modified microbiome in great tits. BMC Evol Biol 14:134–111. https://doi.org/10.1186/1471-2148-14-134
James PJ (1999) Do sheep regulate the size of their mallophagan louse populations? Int J Parasitol 29:869–875. https://doi.org/10.1016/S0020-7519(99)00055-7
Kellogg VL, Kuawana SI (1902) Papers from the Hopkins Stanford Galapagos Expedition, 1898–1899. X. Entomological results (8). Mallophaga from birds. Proc Wash Acad Sci 4:457–499
King MO, Owen JP, Schwabl H (2011) Injecting the mite into ecological immunology: measuring the antibody response of house sparrows (Passer domesticus) challenged with hematophagous mites. Auk 128:340–345. https://doi.org/10.1525/auk.2011.10253
Klein J, Sato A, Nikolaidis N (2007) MHC, TSP, and the origin of species: from immunogenetics to evolutionary genetics. Annu Rev Genet 41:281–304. https://doi.org/10.1146/annurev.genet.41.110306.130137
Koop JAH, Clayton DH (2013) Evaluation of two methods for quantifying passeriform lice. J Field Ornithol 84:210–215. https://doi.org/10.1111/jofo.12020
Leclaire S, van Dongen WFD, Voccia S, Merkling T, Ducamp C, Hatch SA, Blanchard P, Danchin É, Wagner RH (2015) Preen secretions encode information on MHC similarity in certain sex-dyads in a monogamous seabird. Sci Rep 4:6920–6926. https://doi.org/10.1038/srep06920
Leclaire S, Strandh M, Dell’Ariccia G et al (2019) Plumage microbiota covaries with the major histocompatibility complex in blue petrels. Mol Ecol 28:833–846. https://doi.org/10.1111/mec.14993
Lehmann T (1993) Ectoparasites: direct impact on host fitness. Parasitol Today 9:13–17. https://doi.org/10.1016/0169-4758(93)90154-8
Lighten J, Papadopulos AST, Mohammed RS, Ward BJ, Paterson I, Baillie L, Bradbury IR, Hendry AP, Bentzen P, van Oosterhout C (2017) Evolutionary genetics of immunological supertypes reveals two faces of the Red Queen. Nat Commun 8:1294–1211. https://doi.org/10.1038/s41467-017-01183-2
Manly KF (2005) Reliability of statistical associations between genes and disease. Immunogenetics 57:549–558. https://doi.org/10.1007/s00251-005-0025-x
Marmesat E, Schmidt K, Saveljev AP, Seryodkin IV, Godoy JA (2017) Retention of functional variation despite extreme genomic erosion: MHC allelic repertoires in the Lynx genus. BMC Evol Biol 17:158. https://doi.org/10.1186/s12862-017-1006-z
Marshall AG (1981) The ecology of ectoparasitic insects. Academic Press, London
Medzhitov R (2007) Recognition of microorganisms and activation of the immune response. Nature 449:819–826. https://doi.org/10.1038/nature06246
Møller AP, Rózsa L (2005) Parasite biodiversity and host defenses: chewing lice and immune response of their avian hosts. Oecologia 142:169–176. https://doi.org/10.1007/s00442-004-1735-8
Møller AP, Erritzøe J, Rózsa L (2010) Ectoparasites, uropygial glands and hatching success in birds. Oecologia 163:303–311. https://doi.org/10.1007/s00442-009-1548-x
Moreno-Rueda G (2010) Uropygial gland size correlates with feather holes, body condition and wingbar size in the house sparrow Passer domesticus. J Avian Biol 41:229–236. https://doi.org/10.1111/j.1600-048X.2009.04859.x
Murphy KP, Janeway C (2008) Janeway’s immunobiology. Garland Science, London
Nováková E, Hypša V, Moran NA (2009) Arsenophonus, an emerging clade of intracellular symbionts with a broad host distribution. BMC Microbiol 9:1–14. https://doi.org/10.1186/1471-2180-9-143
Oliver MK, Telfer S, Piertney SB (2009) Major histocompatibility complex (MHC) heterozygote superiority to natural multi-parasite infections in the water vole (Arvicola terrestris). Proc Biol Sci 276:1119–1128. https://doi.org/10.1098/rspb.2008.1525
Olsson M, Wapstra E, Madsen T, Ujvari B, Rugfelt C (2005) Costly parasite resistance: a genotype-dependent handicap in sand lizards? Biol Lett 1:375–377. https://doi.org/10.1098/rsbl.2005.0339
Oppelt C, Starkloff A, Rausch P et al (2010) Major histocompatibility complex variation and age-specific endoparasite load in subadult European rabbits. Mol Ecol 19:4155–4167. https://doi.org/10.1111/j.1365-294X.2010.04766.x
Owen JP, Delany ME, Mullens B a. (2008) MHC haplotype involvement in avian resistance to an ectoparasite. Immunogenetics 60:621–631
Owen JP, Delany ME, Cardona CJ et al (2009) Host inflammatory response governs fitness in an avian ectoparasite, the northern fowl mite (Ornithonyssus sylviarum). Int J Parasitol 39:789–799. https://doi.org/10.1016/j.ijpara.2008.12.008
Owen JP, Nelson AC, Clayton DH (2010) Ecological immunology of bird-ectoparasite systems. Trends Parasitol 26:530–539. https://doi.org/10.1016/j.pt.2010.06.005
Palma R, Price R (2010) The species of Myrsidea Waterston (Insecta: Phthiraptera: Menoponidae) from the Galápagos Islands, with descriptions of new taxa. Tuhinga 21:135–146
Pilosof S, Fortuna MA, Cosson J-F et al (2014) Host-parasite network structure is associated with community-level immunogenetic diversity. Nat Commun 5:5172–5179. https://doi.org/10.1038/ncomms6172
R Core Team (2015) R: a language and environment for statistical computing
Richner H, Oppliger A, Christe P (1993) Effect of an ectoparasite on reproduction in great tits. J Anim Ecol 62:703. https://doi.org/10.2307/5390
Rock KL, Reits E, Neefjes J (2016) Present yourself! By MHC class I and MHC class II molecules. Trends Immunol 37:724–737. https://doi.org/10.1016/j.it.2016.08.010
Savage AE, Zamudio KR (2011) MHC genotypes associate with resistance to a frog-killing fungus. Proc Natl Acad Sci U S A 108:16705–16710. https://doi.org/10.1073/pnas.1106893108
Schad J, Dechmann DKN, Voigt CC, Sommer S (2012) Evidence for the “good genes” model: association of MHC class II DRB alleles with ectoparasitism and reproductive state in the neotropical lesser bulldog bat, Noctilio albiventris. PLoS One 7:e37101. https://doi.org/10.1371/journal.pone.0037101
Schneider DS, Ayres JS (2008) Two ways to survive infection: what resistance and tolerance can teach us about treating infectious diseases. Nat Rev Immunol 8:889–895. https://doi.org/10.1038/nri2432
Schwensow N, Fietz J, Dausmann KH, Sommer S (2007) Neutral versus adaptive genetic variation in parasite resistance: importance of major histocompatibility complex supertypes in a free-ranging primate. Heredity (Edinb) 99:265–277. https://doi.org/10.1038/sj.hdy.6800993
Seifertová M, Jarkovský J, Šimková A (2016) Does the parasite-mediated selection drive the MHC class IIB diversity in wild populations of European chub (Squalius cephalus)? Parasitol Res 115:1401–1415. https://doi.org/10.1007/s00436-015-4874-4
Sepil I, Lachish S, Hinks AE, Sheldon BC (2013) Mhc supertypes confer both qualitative and quantitative resistance to avian malaria infections in a wild bird population. Proc R Soc B Biol Sci 280:20130134. https://doi.org/10.1098/rspb.2013.0134
Sette A, Sidney J (1998) HLA supertypes and supermotifs: a functional perspective on HLA polymorphism. Curr Opin Immunol 10:478–482. https://doi.org/10.1016/S0952-7915(98)80124-6
Slade R, McCallum H (1992) Overdominant vs. frequency-dependent selection at MHC loci. Genetics 132:861–862
Slade JWG, Watson MJ, Kelly TR et al (2016) Chemical composition of preen wax reflects major histocompatibility complex similarity in songbirds. Proc R Soc B Biol Sci 283:20161966. https://doi.org/10.1098/rspb.2016.1966
Soler JJ, Peralta-Sánchez JM, Martín-Platero AM, Martín-Vivaldi M, Martínez-Bueno M, Møller AP (2012) The evolution of size of the uropygial gland: mutualistic feather mites and uropygial secretion reduce bacterial loads of eggshells and hatching failures of European birds. J Evol Biol 25:1779–1791. https://doi.org/10.1111/j.1420-9101.2012.02561.x
Spurgin LG, Richardson DS (2010) How pathogens drive genetic diversity: MHC, mechanisms and misunderstandings. Proc Biol Sci 277:979–988. https://doi.org/10.1098/rspb.2009.2084
Štefka J, Hoeck PEA, Keller LF, Smith VS (2011) A hitchhikers guide to the Galápagos: cophylogeography of Galápagos mockingbirds and their parasites. BMC Evol Biol 11:284. https://doi.org/10.1186/1471-2148-11-284
Tompkins DM, Mitchell RA, Bryant DM (2006) Hybridization increases measures of innate and cell-mediated immunity in an endangered bird species. J Anim Ecol 75:559–564. https://doi.org/10.1111/j.1365-2656.2006.01076.x
Trachtenberg E, Korber B, Sollars C, Kepler TB, Hraber PT, Hayes E, Funkhouser R, Fugate M, Theiler J, Hsu YS, Kunstman K, Wu S, Phair J, Erlich H, Wolinsky S (2003) Advantage of rare HLA supertype in HIV disease progression. Nat Med 9:928–935. https://doi.org/10.1038/nm893
Untalan PM, Pruett JH, Steelman CD (2007) Association of the bovine leukocyte antigen major histocompatibility complex class II DRB3*4401 allele with host resistance to the lone star tick, Amblyomma americanum. Vet Parasitol 145:190–195. https://doi.org/10.1016/j.vetpar.2006.12.003
Vlček J, Hoeck PEA, Keller LF, Wayhart JP, Dolinová I, Štefka J (2016) Balancing selection and genetic drift create unusual patterns of MHCIIβ variation in Galápagos mockingbirds. Mol Ecol 25:4757–4772. https://doi.org/10.1111/mec.13807
Walker M, Steiner S, Brinkhof MWG, Richner H (2003) Induced responses of nestling great tits reduce hen flea reproduction. Oikos 102:67–74. https://doi.org/10.1034/j.1600-0706.2003.12208.x
Walther BA, Clayton DH (1997) Dust-ruffling: a simple method for quantifying. J Field Ornithol 68:509–518. https://doi.org/10.2307/4514260
Westerdahl H, Asghar M, Hasselquist D, Bensch S (2012) Quantitative disease resistance: to better understand parasite-mediated selection on major histocompatibility complex. Proc Biol Sci 279:577–584. https://doi.org/10.1098/rspb.2011.0917
Westerdahl H, Stjernman M, Råberg L, Lannefors M, Nilsson JÅ (2013) MHC-I affects infection intensity but not infection status with a frequent avian malaria parasite in blue tits. PLoS One 8:e72647. https://doi.org/10.1371/journal.pone.0072647
Whiteman NK, Matson KD, Bollmer JL, Parker PG (2006) Disease ecology in the Galápagos hawk (Buteo galapagoensis): host genetic diversity, parasite load and natural antibodies. Proc Biol Sci 273:797–804. https://doi.org/10.1098/rspb.2005.3396
Wikel SK (1982) Immune responses to arthropods and their products. Annu Rev Entomol 27:21–48. https://doi.org/10.1146/annurev.en.27.010182.000321
Worley K, Collet J, Spurgin LG, Cornwallis C, Pizzari T, Richardson DS (2010) MHC heterozygosity and survival in red junglefowl. Mol Ecol 19:3064–3075. https://doi.org/10.1111/j.1365-294X.2010.04724.x
Acknowledgements
We thank Prof. Lukas Keller and anonymous reviewers for valuable comments on the manuscript. We also thank the Galápagos National Park Service for permission to conduct this research (Permit Nos. PC-48-10 and PC-08-14) and the Charles Darwin Foundation for assistance in the field. We thank Czech Science Foundation and Grant Agency of the University of South Bohemia for funding.
Funding
The research was funded by the Czech Science Foundation (project no. P506/12/P529) and Grant Agency of the University of South Bohemia (project no. 048/2019/P).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Handling Editor: Una Ryan
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vlček, J., Štefka, J. Association between louse abundance and MHC II supertypes in Galápagos mockingbirds. Parasitol Res 119, 1597–1605 (2020). https://doi.org/10.1007/s00436-020-06617-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-020-06617-3