Abstract
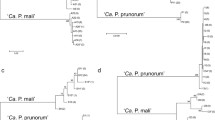
A quarantine organism, “Candidatus Phytoplasma mali,” is the causal agent of apple proliferation, one of the most important apple diseases in Europe. The genetic diversity of this pathogen in Central and Southern Europe has already been reported; however, almost no data exists from Eastern Europe. In this study, “Ca. P. mali” strains, which were identified in 14 apple trees from the Bulgarian germplasm collection, were characterized by restriction fragment length polymorphism (RFLP) and sequence analysis of four genomic loci. In total, nine distinct genetic lineages were recognized based on the combination of the following detected RFLP profiles: two profiles for the 16S-23S rDNA region (16SrX-A2, -A3), four profiles for the secY gene (one previously known: secY(X)-A, and three new: secY-C, secY-D, secY-E), three profiles for the rpl22-rps3 genes (rpX-A, rpX-B, rpX-F), and one profile for the nitroreductase- and rhodanese-like gene (AT-1). Phylogenetic analysis of the Bulgarian and other European “Ca. P. mali” strains based on 16S-23S rRNA gene sequences confirmed RFLP grouping, regardless of the phytoplasma origin. In a phylogenetic tree based on the secY data, only German strains formed separate clade from the other strains. The tree based on rp genes did not correspond to RFLP profiles. Unexpectedly, when using nitroreductase and rhodanese-like gene sequences, the Bulgarian strains clustered separately from the other European strains. Apart from the identification of different “Ca. P. mali” strains, the paper also recommends the unification of the rpX-subgroup nomenclature to avoid future confusions. Both aims of this paper provide valuable tools to understand the epidemiology of this quarantine pathogen.



Similar content being viewed by others
References
Anisimova M, Gascuel O (2006) Approximate likelihood-ratio test for branches: a fast, accurate, and powerful alternative. Syst Biol 55:539–552. https://doi.org/10.1080/10635150600755453
Baric S, Berger J, Cainelli C, Kerschbamer C, Dalla Via J (2011) Molecular typing of “Candidatus Phytoplasma mali” and epidemic history tracing by a combined T-RFLP/VNTR analysis approach. Eur J Plant Pathol 131:573–584. https://doi.org/10.1007/s10658-011-9832-0
Bertaccini A, Paltrinieri S, Martini M, Ermacora P (2008) Comparison of different detection systems for apple proliferation phytoplasmas in Trentino (North Italy). Acta Hort 781:453–458. https://doi.org/10.17660/ActaHortic.2008.781.65
Bertaccini A, Duduk B, Paltrinieri S, Contaldo N (2014) Phytoplasmas and phytoplasma diseases: a severe threat to agriculture. Am J Plant Sci 5:1763–1788. https://doi.org/10.4236/ajps.2014.512191
Borisova A, Borodinova M, Kamenova I (2014) Major diseases of apple trees in Kyustendil region of Bulgaria. Turkish J Agricultur Natur Sci 1:695–700 http://dergipark.gov.tr/download/article-file/142164
Casati P, Quaglino F, Tedeschi R, Spiga FM, Alma A, Spadone P, Bianco PA (2010) Identification and molecular characterization of ‘Candidatus phytoplasma mali’ isolates in north-western Italy. J Phytopathol 158:81–87 http://onlinelibrary.wiley.com/doi/10.1111/j.1439-0434.2009.01581.x/full
Casati P, Quaglino F, Stern AR, Tedeschi R, Alma A, Bianco PA (2011) Multiple gene analyses reveal extensive genetic diversity among “Candidatus Phytoplasma mali” populations. Ann Appl Biol 158:257–266 http://onlinelibrary.wiley.com/doi/10.1111/j.1744-7348.2011.00461.x/full
Cieslinska M, Hennig E, Kruczynska D, Bertaccini A (2015) Genetic diversity of “Candidatus Phytoplasma mali” strains in Poland. Phytopathol Mediterr 54:477–487. https://doi.org/10.14601/Phytopathol_Mediterr-15785
Danet JL, Balakishiyeva G, Cimerman A, Sauvion N, Marie-Jeanne V, Labonne G, Laviňa A, Batlle A, Križanac I, Škorić D, Ermacora P, Serçe CU, Caglayan K, Jarausch W, Foissac X (2011) Multilocus sequence analysis reveals the genetic diversity of European fruit tree phytoplasmas and supports the existence of inter-species recombination. Microbiol 157:438–450. https://doi.org/10.1099/mic.0.043547-0
Davis RE, Lee I-M (1993) Cluster-specific polymerase chain reaction amplification of 16S rDNA sequences for detection and identification of mycoplasmalike organisms. Phytopathology 83:1008–1011 https://www.apsnet.org/publications/phytopathology/backissues/Documents/1993Articles/Phyto83n09_1008.PDF
Dereeper A, Guignon V, Blanc G, Audic S, Buffet S, Chevenet F, Dufayard JF, Guindon S, Lefort V, Lescot M, Claverie JM, Gascuel O (2008) Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res 36:W465–W469. https://doi.org/10.1093/nar/gkn180
Etropolska A (2015) Molecular characterization of fruit tree phytoplasmas and their vectors in Bulgaria. Disertation University of Forestry/AlPlanta Institute for Plant Research, Bulgaria/Germany
Etropolska A, Laginova M (2012) Monitoring of the distribution of fruit tree phytoplasmas in Bulgaria from 2007 until 2011. Petria 22:449–451
Etropolska A, Jarausch W, Jarausch B, Trenchev G (2015) Detection of European fruit tree phytoplasmas and their insect vectors in important fruit-growing regions in Bulgaria. Bulg J Agric Sci 21:1248–1253 http://www.agrojournal.org/21/06-19.pdf
Fránová J, Ludvíková H, Paprštein F, Bertaccini A (2013) Genetic diversity of Czech “Candidatus Phytoplasma mali” strains based on multilocus gene analyses. Eur J Plant Pathol 136:675–688 https://link.springer.com/article/10.1007/s10658-013-0196-5
Frisinghelli C, Delaiti L, Grando M, Forti D, Vindimian M (2000) Cacopsylla costalis (Flor 1861), as a vector of apple proliferation in Trentino. J Phytopathol 148:425–431 https://onlinelibrary.wiley.com/doi/abs/10.1046/j.1439-0434.2000.00403.x
Gundersen DE, Lee I-M (1996) Ultrasensitive detection of phytoplasmas by nested-PCR assays using two universal primer pairs. Phytopathol Mediterr 35:144–151 http://www.jstor.org/stable/42685262
IRPCM (2004) “Candidatus Phytoplasma”, a taxon for the wall-less, non-helical prokaryotes that colonize plant phloem and insects. Int J Syst Evol Microbiol 54:1243–1255. https://doi.org/10.1099/ijs.0.02854-0
Jarausch W, Saillard C, Dosba F, Bove JM (1994) Differentiation of mycoplasmalike organisms (MLOs) in European fruit trees by PCR using specific primers derived from the sequence of a chromosomal fragment of the apple proliferation MLO. Appl Environ Microbiol 60:2916–2923 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC201743/
Jarausch W, Saillard C, Helliot B, Garnier M, Dosba F (2000) Genetic variability of apple proliferation phytoplasmas as determined by PCR-RFLP and sequencing of a non-ribosomal fragment. Mol Cell Probes 14:17–24. https://doi.org/10.1006/mcpr.1999.0279
Jarausch B, Schwind N, Jarausch W, Krczal G, Seemüller E, Dickler E (2003) First report of Cacopsylla picta as a vector for apple proliferation phytoplasma in Germany. Plant Dis 87:101. https://doi.org/10.1094/PDIS.2003.87.1.101A
Lee I-M, Davis RE, Hiruki C (1991) Genetic interrelatedness among clover proliferation mycoplasmalike organisms (MLOs) and other MLOs investigated by nucleic acid hybridization and restriction fragment length polymorphism analyses. Appl Environ Microbiol 57:3565–3569 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC184013/
Lee I-M, Gundersen DE, Hammond RW, Davis RE (1994) Use of mycoplasmalike organism (MLO) group-specific oligonucleotide primers for nested-PCR assays to detect mixed-MLO infections in a single host plant. Phytopathology 84:559–566 https://www.apsnet.org/publications/phytopathology/backissues/Documents/1994Articles/Phyto84n06_559.PDF
Lee I-M, Martini M, Bottner KD, Dane RA, Black MC, Troxclair N (2003) Ecological implications from a molecular analysis of phytoplasmas involved in an aster yellows epidemic in various crops in Texas. Phytopathology 93:1368–1377. https://doi.org/10.1094/PHYTO.2003.93.11.1368
Lee I-M, Gundersen-Rindal DE, Davis RE, Bottner KD, Marcone C, Seeműller E (2004) “Candidatus Phytoplasma asteris”, a novel phytoplasma taxon associated with aster yellows and related diseases. Int J Syst Evol Microbiol 54:1037–1048. https://doi.org/10.1099/ijs.0.02843-0
Letunic I, Bork P (2016) Interactive tree of life (iTOL) v3: an online tool for the display and annotation of phylogenetic and other trees. Nucleic Acids Res 44:W242–W245. https://doi.org/10.1093/nar/gkw290
Martini M, Lee I-M, Bottner KD, Zhao Y, Botti S, Bertaccini A, Harrison NA, Carraro L, Marcone C, Khan AJ, Osler R (2007) Ribosomal protein gene-based phylogeny for finer differentiation and classification of phytoplasmas. Int J Syst Evol Microbiol 57:2037–2051. https://doi.org/10.1099/ijs.0.65013-0
Martini M, Ermacora P, Falginella L, Loi N, Carraro L (2008) Molecular differentiation of “Candidatus Phytoplasma mali” and its spreading in Friuli Venezia Giulia region (North-East Italy). Acta Hort 781:395–402. https://doi.org/10.17660/ActaHortic.2008.781.56
Milusheva S, Terziev I (2013) Preliminary results from investigation on apple proliferation. Acta Hort 981:513–515. https://doi.org/10.17660/ActaHortic.2013.981.82
Padovan AC, Gibb KS, Bertaccini A, Vibio M, Bonfiglioli RE, Magarey PA, Sears BB (1995) Molecular detection of the Australian grapevine yellows phytoplasma and comparison with grapevine yellows phytoplasmas from Italy. Aust J Grape Wine Res 1:25–31 http://onlinelibrary.wiley.com/doi/10.1111/j.1755-0238.1995.tb00074.x/full
Paltrinieri S, Duduk B, Dal Molin F, Comerlati G, Bertaccini A (2010) Molecular characterization of “Candidatus Phytoplasma mali” strains in outbreaks of apple proliferation in north eastern Italy, Hungary, and Serbia. Julius-Kühn-Archiv 427:178–182 https://ojs.openagrar.de/index.php/JKA/article/view/431/1483
Rui D, Ciferri R, Refatti E (1950) La virosi degli “scopazzi del melo” nel Veronese. Not Mal Piante 13:7–11
Seemüller E, Schneider B (2004) “Candidatus Phytoplasma mali”, “Candidatus Phytoplasma pyri” and “Candidatus Phytoplasma prunorum”, the causal agents of apple proliferation, pear decline and European stone fruit yellows, respectively. Int J Syst Evol Microbiol 54:1217–1226. https://doi.org/10.1099/ijs.0.02823-0
Seemüller E, Kiss E, Sule S, Schneider B (2010) Multiple infection of apple trees by distinct strains of “Candidatus Phytoplasma mali” and its pathological relevance. Phytopathology 100:863–870. https://doi.org/10.1094/PHYTO-100-9-0863
Tedeschi R, Alma A (2004) Transmission of apple proliferation phytoplasma by Cacopsylla melanoneura (Homoptera: Psyllidae). J Econ Entomol 97:8–13. https://doi.org/10.1093/jee/97.1.8
Trifonov D (1965) Apple proliferation, witches’ broom. Spread and damage. Hort Vitic Sci 2:437–444
Trifonov D (1975) Apple proliferation witches’ broom. Experimental evidence of the spread of the disease in nature. Plant Protect Sci 1–2:29–33
Valasevich N, Schneider B (2016) Detection, identification, and characterization of phytoplasmas infecting apple and pear trees in Belarus. Plant Dis 100:2275–2280 https://apsjournals.apsnet.org/doi/pdf/10.1094/PDIS-04-16-0498-RE
Acknowledgements
The authors thank Prof. A. Bertaccini and Mrs. S. Paltrinieri (University of Bologna, Italy), Prof. S. Barić (Free University of Bozen – Bolzano, Italy), and Mrs. S. Öttl (Research Centre Laimburg, Italy) for kindly providing the phytoplasma positive controls. The authors are deeply indebted to Mrs. J. Rakouská and O. Baudysová for their technical assistance.
Funding
This research was supported by the grants from the Ministry of Education, Youth and Sports of the Czech Republic No. LD12074, and the institutional support RVO60077344 from the Czech Academy of Sciences
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 1029 kb)
Rights and permissions
About this article
Cite this article
Fránová, J., Koloniuk, I., Lenz, O. et al. Molecular diversity of “Candidatus Phytoplasma mali” strains associated with apple proliferation disease in Bulgarian germplasm collection. Folia Microbiol 64, 373–382 (2019). https://doi.org/10.1007/s12223-018-0660-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-018-0660-x