Abstract
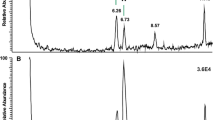
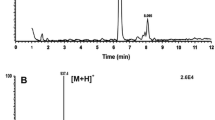
The rationale of “green pesticides” in food security is to use information about endogenous hormones of pest insects to make peptide mimetics that will act against the pest insects to alter their behaviour or physiology, while taking care not to harm beneficial insects or other organisms in the food chain. Such “green” insecticides are designed thus, on the basis of neuropeptide ligand–receptor interaction and it is of paramount interest to have finally a mimetic at hand that is harmful only to pest insects. For this concept to work, one has to identify the ligands in pest and beneficial insects. In this study we investigate adipokinetic hormones (AKHs) from a hemipteran source. The most harmful hemipterans on an economic scale are aphids (Hemiptera: Sternorrhyncha: Aphidoidea) of which the AKH is known. Here we identify the AKH complement of a member of a related suborder, the raintree bug or froghopper Ptyelus flavescens (Hemiptera: Cicadomorpha: Cercopoidea). Identification and sequence elucidation of the adipokinetic peptides of this species was achieved by a heterospecific and conspecific trehalose-mobilizing bioassay, and by liquid chromatography coupled to positive electrospray mass spectrometry (LC–ESI–MS) including tandem MS2 spectra obtained by collision-induced dissociation. High resolution MS was employed to distinguish between Gln and Lys residues in the peptides. Three AKHs are discovered in the raintree bug: an octapeptide (Peram-CAH-I: pEVNFSPNW amide) previously known from cockroaches, and two novel decapeptides (Ptyfl-AKH-I: pEINFSTGWGQ amide and Ptyfl-AKH-II: pEINFSTAWGQ amide). The novel peptides were synthesized and the sequence assignments were unequivocally confirmed by co-elution of synthetic peptides and the natural equivalent and by identical MS data of the two forms. A conspecific bioassay in the froghopper describe the endogenous peptide Ptyfl-AKH-I as hypertrehalosemic. In heterologous bioassays the two novel AKHs induce an increase of circulating carbohydrates in cockroaches: Ptyfl-AKH-I is much more active than Ptyfl-AKH-II. Moreover, if the Ile2 in Ptyfl-AKH-II is replaced with a Leu2 residue, biological activity is further diminished. The current data show that the raintree AKH decapeptides differ by four amino acids from the aphid AKH (Acypi-AKH: pEVNFTPTWGQ amide). Therefore, it may be permissible to use the aphid AKH ligand–receptor pair to develop a “green” insecticide to target aphid metabolism.





Similar content being viewed by others
References
Altstein M, Nässel DR (2010) Neuropeptide signalling in insects. In: Geary TG, Maule AG (eds) Neuropeptide systems as targets for parasite and pest control (Chapter 8). Landes Bioscience and Springer Science+Business Media, Austin and Berlin
Audsley N, Down RE (2015) G protein coupled receptors as targets for next generation pesticides. Insect Biochem Mol Biol 67:27–37
Caers J, Peeters L, Janssen T et al (2012) Structure-activity studies of Drosophila adipokinetic hormone (AKH) by a cellular expression system of dipteran AKH receptors. Gen Comp Endocrinol 177:332–337
Chipman AD, Ferrier DEK, Brena C et al (2014) The first myriapod genome sequence reveals conservative arthropod gene content and genome organisation in the centipede Strigamia maritima. PLoS Biol 12:e1002005
Derst C, Dircksen H, Meusemann K et al (2016) Evolution of neuropeptides in non-pterygote hexapods. BMC Evol Biol 16:51
Forero D (2008) The systematics of the Hemiptera. Rev Colombiana Entomol 34:1–21
Gäde G (1980) Further characteristics of adipokinetic and hyperglycaemic factor(s) of stick insects. J Insect Physiol 26:351–360
Gäde G (1992) The hormonal integration of insect flight metabolism. Zool Jb Physiol 96:211–225
Gäde G (2004) Regulation of intermediary metabolism and water balance of insects by neuropeptides. Ann Rev Entomol 49:93–113
Gäde G (2009) Peptides of the adipokinetic hormone/red pigment-concentrating hormone family. A new take on biodiversity. Ann NY Acad Sci 1163:125–136
Gäde G, Goldsworthy GJ (2003) Insect peptide hormones: a selective review of their physiology and potential application for pest control. Pest Manag Sci 59:1063–1075
Gäde G, Janssens MP-E (1994) Cicadas contain novel members of the AKH/RPCH family peptides with hypertrehalosaemic activity. Biol Chem Hoppe Seyler 375:803–809
Gäde G, Marco HG (2009) Peptides of the adipokinetic hormone/red pigment-concentrating hormone family with special emphasis on Caelifera: primary sequences and functional considerations contrasting grasshoppers and locusts. Gen Comp Endocrinol 162:59–68
Gäde G, Marco HG (2013) AKH/RPCH peptides. In: Kastin AJ (ed) Handbook of biologically active peptides (Chapter 28). Elsevier, Amsterdam, pp 185–190
Gäde G, Marco HG (2017) The adipokinetic hormone of Mantodea in comparison to other Dictyoptera. Arch Insect Biochem Physiol 94:e21376
Gäde G, Goldsworthy GJ, Kegel G et al (1984) Single step purification of locust adipokinetic hormones I and II by reversed phase high-performance liquid chromatography and the amino-acid composition of the hormone II. Hoppe Seylers Z Physiol Chem 365:393–398
Gäde G, Auerswald L, Šimek P et al (2003) Red pigment-concentrating hormone is not limited to crustaceans. Biochem Biophys Res Commun 309:972–978
Gäde G, Auerswald L, Predel R et al (2004) Substrate usage and its regulation during flight and swimming in the backswimmer, Notonecta glauca. Physiol Entomol 29:84–93
Gäde G, Šimek P, Marco HG (2007a) Water scorpions (Heteroptera, Nepidae) and giant water bugs (Heteroptera, Belostomatidae): sources of new members of the adipokinetic hormone/red pigment-concentrating hormone family. Peptides 28:1359–1367
Gäde G, Šimek P, Marco HG (2007b) A novel adipokinetic peptide in a water boatman (Heteroptera, Corixidae) and its bioanalogue in a saucer bug (Heteroptera, Naucoridae). Peptides 28:594–601
Gäde G, Šimek P, Marco HG (2015) Two novel tyrosine-containing peptides (Tyr4) of the adipokinetic hormone family in beetles of the families Coccinellidae and Silphidae. Amino Acids 47:2323–2333
Gäde G, Šimek P, Marco HG (2016) Novel members of the adipokinetic hormone family in beetles of the superfamily Scarabaeoidea. Amino Acids 48:2785–2798
Gullan PJ, Cranston PS (2014) The insects: an outline of entomology, 5th edn. Wiley-Blackwell, Chichester
Hayes TK, Keeley LL, Knight DW (1986) Insect hypertrehalosaemic hormone: isolation and primary structure from Blaberus discoidalis cockroaches. Biochem Biophys Res Commun 140:674–678
Holwerda DA, van Doorn J, Beenakkers AMT (1977) Characterization of the adipokinetic and hyperglycaemic substances from the locust corpus cardiacum. Insect Biochem 7:151–157
Jedlička P, Steinbauerová V, Šimek P et al (2012) Functional characterization of the adipokinetic hormone in the pea aphid, Acyrthosiphon pisum. Comp Biochem Physiol A Mol Integr Physiol 162:51–58
Jedličková V, Jedlička P, Lee HJ (2015) Characterization and expression analysis of adipokinetic hormone and its receptor in eusocial aphid Pseudoregma bambucicola. Gen Comp Endocrinol 223:38–46
Johnson JI, Kavanaugh SI, Nguyen C et al (2014) Localization and functional characterization of a novel adipokinetic hormone in the mollusc, Aplysia californica. PloS One 9:e106014
Kodrík D, Marco HG, Šimek P et al (2010) The adipokinetic hormones of Heteroptera: a comparative study. Physiol Entomol 35:117–127
Li S, Hauser F, Skadborg SK et al (2016) Adipokinetic hormones and their G protein-coupled receptors emerged in Lophotrochozoa. Sci Rep 6:32789
Lorenz MW, Gäde G (2009) Hormonal regulation of energy metabolism in insects as a driving force for performance. Integr Comp Biol 49:380–392
Lorenz MW, Kellner R, Völkl W et al (2001) A comparative study on hypertrehalosaemic hormones in the Hymenoptera: sequence determination, physiological actions and biological significance. J Insect Physiol 47:563–571
Nygaard S, Zhang G, Schiøtt M et al (2011) The genome of the leaf-cutting ant Acromyrmex echinatior suggests key adaptations to advanced social life and fungus farming. Genome Res 21:1339–1348
Spik G, Montreuil J (1964) Deux causes d‘erreur dans les dosages colorimetriques des oses neutres totaux. Bull Soc Chim Biol 46:739–749
The International Aphid Genomics Consortium (2010) Genome sequence of the pea aphid Acyrthosiphon pisum. PLoS Biol 8:e1000313
Veenstra JA, Rodriguez L, Weaver RJ (2012) Allatotropin, leucokinin and AKH in honey bees and other Hymenoptera. Peptides 35:122–130
Verlinden H, Vleugels R, Zels S et al (2015) Receptors for neuronal or endocrine signalling molecules as potential targets for the control of insect pests. Adv Insect Physiol 46:167–303
Weaver RJ, Marco HG, Šimek P et al (2012) Adipokinetic hormones of sphingid Lepidoptera, including the identification of a second M. sexta AKH. Peptides 34:44–50
Wenger JA, Cassone BJ, Legeai F et al (2017) Whole genome sequence of the soybean aphid, Aphis glycines. Insect Biochem Mol Biol. doi:10.1016/j.ibmb.2017.01.005
Xu G, Gu G-X, Teng Z-W et al (2016) Identification and expression profiles of neuropeptides and their G protein-coupled receptors in the rice stem borer Chilo suppressalis. Sci Rep 6:28976
Zöllner N, Kirsch K (1962) Über die quantitative Bestimmung von Lipoiden (Mikromethode) mittels der vielen natürlichen Lipoiden (allen bekannten Plasmalipoiden) gemeinsamen Sulfophosphovanillin-Reaktion. Z Gesamte Exp Med 135:545–561
Acknowledgements
This work is based on the research supported in part by the National Research Foundation of South Africa (Grant No 85768 [IFR13020116790 to GG] and Grant No. 10924 to HGM). The authors also thank the following organisations for financial support: the Research Council of the University of Cape Town (staff awards to GG and HGM) and the Czech Science Foundation (Project No. 17-22276S to PS). We thank P. Krusberska and M. Moos for help with the MS experiments, and Vic Le Roux for his help with collecting the raintree bugs.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Handling Editor: M. S. Palma.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gäde, G., Šimek, P. & Marco, H.G. The African froghopper Ptyelus flavescens (suborder: Cicadomorpha) contains two novel and one known peptides of the adipokinetic hormone (AKH) family: structure, function and comparison with aphid AKH (suborder: Sternorrhyncha). Amino Acids 49, 1679–1690 (2017). https://doi.org/10.1007/s00726-017-2461-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-017-2461-y