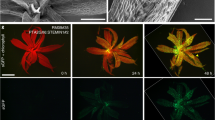
Abstract
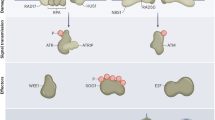
DNA damage can result from intrinsic cellular processes and from exposure to stressful environments. Such DNA damage generally threatens genome integrity and cell viability1. However, here we report that the transient induction of DNA strand breaks (single-strand breaks, double-strand breaks or both) in the moss Physcomitrella patens can trigger the reprogramming of differentiated leaf cells into stem cells without cell death. After intact leafy shoots (gametophores) were exposed to zeocin, an inducer of DNA strand breaks, the STEM CELL-INDUCING FACTOR 1 (STEMIN1)2 promoter was activated in some leaf cells. These cells subsequently initiated tip growth and underwent asymmetric cell divisions to form chloronema apical stem cells, which are in an earlier phase of the life cycle than leaf cells and have the ability to form new gametophores. This DNA-strand-break-induced reprogramming required the DNA damage sensor ATR kinase, but not ATM kinase, together with STEMIN1 and closely related proteins. ATR was also indispensable for the induction of STEMIN1 by DNA strand breaks. Our findings indicate that DNA strand breaks, which are usually considered to pose a severe threat to cells, trigger cellular reprogramming towards stem cells via the activity of ATR and STEMINs.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The sequence data can be found in Phytozome P. patens v.3.3 (ref. 45) (OE-JGI, http://phytozome.jgi.doe.gov/) under the following accession numbers: ATM (Pp3c2_23700), ATR (Pp3c6_3460), STEMIN1 (Pp3c1_27440), STEMIN2 (Pp3c14_9940), STEMIN3 (Pp3c10_7030), CSP1 (Pp3c5_6070), CSP2 (Pp3c6_23240), CSP3 (Pp3c5_7920) and CSP4 (Pp3c5_7880). RNA-seq data were deposited into the DDBJ Sequence Read Archive (DRA) under accession number DRA008745.
References
Yoshiyama, K. O., Sakaguchi, K. & Kimura, S. DNA damage response in plants: conserved and variable response compared to animals. Biology 2, 1338–1356 (2013).
Ishikawa, M. et al. Physcomitrella STEMIN transcription factor induces stem cell formation with epigenetic reprogramming. Nat. Plants 5, 681–690 (2019).
Ikeuchi, M., Ogawa, Y., Iwase, A. & Sugimoto, K. Plant regeneration: cellular origins and molecular mechanisms. Development 143, 1442–1451 (2016).
Pilkington, M. The regeneration of the stem apex. New Phytol. 28, 37–53 (1929).
Reinhardt, D., Frenz, M., Mandel, T. & Kuhlemeier, C. Microsurgical and laser ablation analysis of interactions between the zones and layers of the tomato shoot apical meristem. Development 130, 4073–4083 (2003).
Heyman, J. et al. ERF115 controls root quiescent center cell division and stem cell replenishment. Science 342, 860–863 (2013).
Sena, G., Wang, X., Liu, H. Y., Hofhuis, H. & Birnbaum, K. D. Organ regeneration does not require a functional stem cell niche in plants. Nature 457, 1150–1153 (2009).
Zhou, W. et al. A Jasmonate signaling network activates root stem cells and promotes regeneration. Cell 177, 942–956 (2019).
Fulcher, N. & Sablowski, R. Hypersensitivity to DNA damage in plant stem cell niches. Proc. Natl Acad. Sci. USA 106, 20984–20988 (2009).
Johnson, R. A. et al. SUPPRESSOR OF GAMMA RESPONSE 1 links DNA damage response to organ regeneration. Plant Physiol. 176, 1665–1675 (2017).
Steward, F. C., Mapes, M. O. & Mears, K. Growth and organized development of cultured cells. II. Organization in cultures grown from freely suspended cells. Am. J. Bot. 45, 705–709 (1958).
Kareem, A. et al. PLETHORA genes control regeneration by a two-step mechanism. Curr. Biol. 25, 1017–1030 (2015).
Ishikawa, M. et al. Physcomitrella cyclin-dependent kinase A links cell cycle reactivation to other cellular changes during reprogramming of leaf cells. Plant Cell 23, 2924–2938 (2011).
Sato, Y. et al. Cells reprogramming to stem cells inhibit the reprogramming of adjacent cells in the moss Physcomitrella patens. Sci. Rep. 7, 1909 (2017).
Li, C. et al. A Lin28 homolog reprograms differentiated cells to stem cells in the moss Physcomitrella patens. Nat. Commun. 8, 14242 (2017).
Kofuji, R. & Hasebe, M. Eight types of stem cells in the life cycle of the moss Physcomitrella patens. Curr. Opin. Plant Biol. 17, 13–21 (2014).
Chankova, S. G., Dimova, E., Dimitrova, M. & Bryant, P. E. Induction of DNA double-strand breaks by zeocin in Chlamydomonas reinhardtii and the role of increased DNA double-strand breaks rejoining in the formation of an adaptive response. Radiat. Environ. Biophys. 46, 409–416 (2007).
Chen, J., Ghorai, M. K., Kenney, G. & Stubbe, J. A. Mechanistic studies on bleomycin-mediated DNA damage: multiple binding modes can result in double-stranded DNA cleavage. Nucleic Acids Res. 36, 3781–3790 (2008).
Sakasai, R. & Iwabuchi, K. The distinctive cellular responses to DNA strand breaks caused by a DNA topoisomerase I poison in conjunction with DNA replication and RNA transcription. Genes Genet. Syst. 90, 187–194 (2015).
Angelis, K. J., Dušinská, M. & Collins, A. R. Single cell gel electrophoresis: detection of DNA damage at different levels of sensitivity. Electrophoresis 20, 2133–2138 (1999).
Lanier, C., Manier, N., Cuny, D. & Deram, A. The comet assay in higher terrestrial plant model: review and evolutionary trends. Environ. Pollut. 207, 6–20 (2015).
Jones, K., Kim, D. W., Park, J. S. & Khang, C. H. Live-cell fluorescence imaging to investigate the dynamics of plant cell death during infection by the rice blast fungus Magnaporthe oryzae. BMC Plant Biol. 16, 69 (2016).
Antosiewicz-Bourget, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007).
Hu, Z., Cools, T. & De Veylder, L. Mechanisms used by plants to cope with DNA damage. Annu. Rev. Plant Biol. 67, 439–462 (2016).
Collonnier, C. et al. CRISPR–Cas9-mediated efficient directed mutagenesis and RAD51-dependent and RAD51-independent gene targeting in the moss Physcomitrella patens. Plant Biotechnol. J. 15, 122–131 (2017).
Hong, J. H. et al. A sacrifice-for-survival mechanism protects root stem cell niche from chilling stress. Cell 170, 102–113 (2017).
Sharma, P., Jha, A. B., Dubey, R. S. & Pessarakli, M. Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J. Bot. 2012, 217037 (2012).
Rodriguez, E., Azevedo, R., Fernandes, P. & Santos, C. Cr(VI) induces DNA damage, cell cycle arrest and polyploidization: a flow cytometric and comet assay study in Pisum sativum. Chem. Res. Toxicol. 24, 1040–1047 (2011).
Gehring, M., Reik, W. & Henikoff, S. DNA demethylation by DNA repair. Trends Genet. 25, 82–90 (2009).
Gursoy-Yuzugullu, O., House, N. & Price, B. D. Patching broken DNA: nucleosome dynamics and the repair of DNA breaks. J. Mol. Biol. 428, 1846–1860 (2016).
Rensing, S. A. et al. The Physcomitrella genome reveals evolutionary insights into the conquest of land by plants. Science 319, 64–69 (2008).
Nishiyama, T., Hiwatashi, Y., Sakakibara, K., Kato, M. & Hasebe, M. Tagged mutagenesis and gene-trap in the moss, Physcomitrella patens by shuttle mutagenesis. DNA Res. 7, 9–17 (2000).
Aoyama, T. et al. AP2-type transcription factors determine stem cell identity in the moss Physcomitrella patens. Development 139, 3120–3129 (2012).
Enciso, M., Sarasa, J., Agarwal, A., Fernández, J. L. & Gosálvez, J. A two-tailed comet assay for assessing DNA damage in spermatozoa. Reprod. Biomed. Online 18, 609–616 (2009).
Cortés-Gutiérrez, E. I., Fernández, J. L., Dávila-Rodríguez, M. I., López-Fernández, C. & Gosálvez, J. Two-tailed comet assay (2T-Comet): simultaneous detection of DNA single and double strand breaks. Methods Mol. Biol. 1560, 285–293 (2017).
Gyori, B. M., Venkatachalam, G., Thiagarajan, P. S., Hsu, D. & Clement, M. V. OpenComet: an automated tool for comet assay image analysis. Redox Biol. 2, 457–465 (2014).
R: A Language and Environment for Statistical Computing v.3.5.1 (R Core Team, 2018); https://doi.org/10.1787/csp-aut-table-2018-1-en
Wickham, H. ggplot2: Elegant graphics for data analysis v.3.2.1 https://doi.org/10.1007/978-3-319-24277-4_2 (2016).
Odell, J., Caimi, P., Sauer, B. & Russell, S. Site-directed recombination in the genome of transgenic tobacco. Mol. Gen. Genet. 223, 369–378 (1990).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17, 10–12 (2011).
Bray, N. L., Pimentel, H., Melsted, P. & Pachter, L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 34, 525–527 (2016).
Pimentel, H., Bray, N. L., Puente, S., Melsted, P. & Pachter, L. Differential analysis of RNA-seq incorporating quantification uncertainty. Nat. Methods 14, 687–690 (2017).
Jari, O. et al. vegan: Community ecology package. R package v.2.5-2 (2018).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25, 402–408 (2001).
Lang, D. et al. The Physcomitrella patens chromosome-scale assembly reveals moss genome structure and evolution. Plant J. 93, 515–533 (2018).
Acknowledgements
We thank F. Nogué for providing the CRISPR–Cas9-related vectors; Y. Horiuchi for modifying the sgRNA expression vector; S. Ooi, E. Aoki, M. Goto, T. Nishi, I. Kajikawa and T. Masuoka for technical assistance; and B. Scheres for comments on the draft. The moss cultivation and RNA-seq were supported in part by the Model Plant Research Facility, Functional Genomics Facility, and Data Integration and Analysis Facility at the National Institute for Basic Biology, Japan. N.G. was supported by the RA program of the National Institute for Basic Biology, a scholarship from the China Scholarship Council (no. 201506760024) and a “Double World-classes” development scholarship from Huazhong Agricultural University, the College of Life Science and Technology. This work was partially supported by JSPS KAKENHI grant nos. JP16K14760, JP18H04790 and JP19H05274 to Y.T., 18H04846 to M.I., and 16H06378 to Y.T., M.I. and M.H.; the Young Researcher Overseas Visits Program for Vitalizing Brain Circulation to A.I., M.I., Y.T. and M.H.; the NIBB Collaborative Research Program (grant nos. 18-345 and 19-313), the National Natural Science Foundation of China (grant no. 31672112), Fundamental Research Funds for the Central Universities in China (grant no. 2662018PY099) and the Advanced Foreign Experts Project (grant no. G20190017014) to C.C.; and the Ministry of Education, Youth, and Sports of the Czech Republic grant no. LTC17047/2017 to K.J.A.
Author information
Authors and Affiliations
Contributions
N.G., Y.T., A.I., C.C. and M.H. conceived and designed the research. N.G. and Y.T. performed the experiments. S.S. performed the mRNA sequencing. G.P. analysed the RNA-seq data. Y.K. performed the transformations. A.I. and K.J.A. contributed to the comet assay. M.I. provided mutant lines of STEMINs. N.G. prepared the figures. N.G. and Y.T. wrote the manuscript with contributions from M.I., C.C. and M.H.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Plants thanks Anne Britt, Robert Sablowski and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
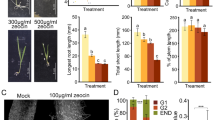
Extended Data Fig. 1 DNA strand break-inducing reagents attenuate the growth of protonemata.
a A representative wild-type leaf at 0, 12, 24, 36, 48, 60, and 72 hours after excision. Blue asterisks indicate the positions of chloronema apical stem cells generated from the edge cells of the excised leaf. The reprogramming of excised leaves was observed in more than two independent experiments with similar results. b–d Representative one-week-old wild-type plants on solid BCDAT medium with or without various concentrations of zeocin (b), bleomycin (BLM) (c), or camptothecin (CPT) (d). Protonema growth slowed under the treatment with 0.5 µg/mL zeocin, 0.1 µg/mL BLM, or 200 nM CPT. Three plants in each concentration of zeocin, BLM, or CPT were observed twice independently with similar results. Scale bars: 100 µm in (a), 1 mm in (b–d).
Extended Data Fig. 2 Newly formed gametophores in protonemata regenerated from intact gametophore leaves after zeocin treatment.
a–f Top panels connected by gray arrows show the life cycle of Physcomitrella. A representative image at each developmental stage is shown. d, g–i Right-bottom panels connected by blue arrows show the regeneration process of excised leaves. Red arrow in (i) indicates a young gametophore formed from protonemata (essentially the same as shown in [b]), which were generated from excised leaves. These new gametophores frequently developed near the original excised leaf. d, j and k Left-bottom panels connected by green arrows show the regeneration process from intact gametophores after temporary zeocin treatment. Wild-type intact gametophores were treated with 50 µg/ml zeocin for 6 hours and incubated in BCDAT liquid medium without a DNA damage reagent for an additional 2 weeks. Orange asterisks in (j) indicate the positions of chloronema apical stem cells generated from differentiated gametophore leaf cells. Red arrows in (k) indicate young gametophores formed from protonemata (essentially the same as shown in [b]) generated from leaves. These newly formed gametophores developed near the leaf. The growth of protonema cells was attenuated in liquid medium. Similar results were obtained using 20 to 25 gametophores in three independent experiments. Scale bars: 20 µm in (a); 100 µm in (b, c, e, f); 1 mm in (d, j, k); 200 µm in (g–i).
Extended Data Fig. 3 Protrusion of chloronema apical stem cells from leaf cells after temporary zeocin treatment.
Representative leaf of an intact gametophore of the H2B-mRFP14 line, with nuclei labeled with histone H2B-mRFP fusion proteins, after 6 hours of 50 µg/ml zeocin (top panels) or mock (bottom panels) treatment. Bright-field (BF) and fluorescence images (mRFP) were collected at 2-hour intervals; images taken at 12, 24, 48, 72, and 78 hours after the initiation of treatment are shown. Images taken at 24, 48, and 72 hours are 1.75× magnified views of the regions highlighted by red squares in the images taken at 12 and 78 hours. Red arrows indicate leaf cells that were ultimately reprogrammed into chloronema stem cells. The experiments were performed twice independently with similar results. Scale bars: 200 µm.
Extended Data Fig. 4 Reprogramming induced by transient zeocin treatment occurs independently of dead cells.
a Representative leaves without (top panels) or with visible dead cells (bottom panels) after transient zeocin or BLM treatment for 6 hours, followed by 7 days of cultivation without DNA damage reagents. Red arrows indicate dead cells in brown. b Percentage of gametophores with at least one visible dead leaf cell. Dark red dots and bars represent the mean and SD from three independent experiments (30 ≤ n ≤ 42, gametophores), respectively. c Bright-field (BF) and PI fluorescence images (PI) of a representative gametophore leaf without dead cells during and after 50 µg/ml zeocin treatment for 6 hours. Images taken at 0, 6 (just before washing out zeocin), 12, 24, 36, 48, 60, 72, and 84 h from the time-lapse images (Supplementary Video 1) are shown. Yellow arrows indicate reprogrammed cells with protrusion. Although PI does not penetrate into apoplastic space of intact gametophore tissue, staining of cell wall with PI became visible after 64 h (Supplementary Video 1). After protrusion started in reprogrammed cells, PI fluorescence in cell walls of surrounding cells became visible, likely because of higher penetration in reprogrammed cells with tip growth. Similar results have been observed in 6 different gametophores. Since only the intercellular space of surrounding cells were stained by PI and none of the cells show nucleus staining, no imperceptible dead cells existed around the protruding reprogrammed cells to trigger the reprogramming. d Bright-field (BF) and PI fluorescence images (PI) of a representative gametophore leaf with dead cells during and after 50 µg/ml zeocin treatment for 6 hours. Images were taken from the time-lapse images (Supplementary Video 2) at the same timepoints as those in (c). Yellow and pink arrows indicate reprogrammed cells with protrusion and dead cells, respectively. After protrusion started in reprogrammed cells, PI fluorescence in cell walls of surrounding cells became visible, likely because of higher penetration in reprogrammed cells with tip growth. Also, PI fluorescence was detected in cell walls surrounding dead cells. Similar results have been observed in 2 different gametophores. Scale bars: 100 µm in (a); 100 µm in (c, d).
Extended Data Fig. 5 Induction of CYCD1;1, CSP1, STEMIN1, and closely related genes in reprogramming cells after transient zeocin treatment.
a Representative gametophore leaves of CYCD1;1pro:NGG (NLS-sGFP-GUS) #26313, nPpCSP1-Citrine-3ʹUTR #115, STEMIN1pro:NGG #7, STEMIN2pro:NGG #238, and STEMIN3pro:NGG #262 after 6 hours of 50 µg/ml zeocin treatment. Bright-field images taken at 10 and 72 h and fluorescent images taken at 10, 24, or 25, 44, 64, and 72 h from time-lapse images (Supplementary Videos 3-7) are shown. White arrows in the middle panels indicate chloronema cells near the observed leaf. The experiments were performed twice independently with similar results. Scale bars: 200 µm. b Intensities of sGFP or Citrine signals in three independent protruded and un-protruded cells of intact gametophore leaves of the lines described in (a) after zeocin treatment. Signal intensity was measured with Fiji 1.0. Red arrows indicate time points at which the cells began to protrude.
Extended Data Fig. 6 Neither ATM nor ATR deletions affect reprogramming induced by wounding.
a Representative wild-type, ∆atm, and ∆atr leaves cultured for 3 days after excision. b Percentages of wild-type, ∆atm, and ∆atr leaves with protruding edge cells (left) and non-edge cells (right) cultured for 3 days after excision. Blue and violet dots and bars represent the mean and SD from three independent experiments (n = 32, excised leaves), respectively. Scale bar: 200 µm in (a).
Extended Data Fig. 7 Transcript levels of DNA damage response genes with or without zeocin treatment in ∆atm or ∆atr deletion mutants.
a–k Relative transcript levels of the genes in ∆atm-del #1, ∆atr-del #13, and wild type with (orange dots and lines) or without zeocin treatment (gray dots and lines) for 6 hours detected with RT-qPCR. Primers used in RT-qPCR are shown in Supplementary Table 2. The highest transcript value of each gene was normalized to 1.0. Dots and bars indicate means and SD of the technical triplicates (n = 3). Two biological replicates were analyzed with similar result.
Supplementary information
Supplementary Information
Supplementary Figs. 1–7, Tables 1 and 2, and References 1–4.
41477_2020_745_MOESM3_ESM.avi
Supplementary Video 1: PI fluorescence in leaves without dead cells of an intact gametophore during and after temporary zeocin treatment. Intact wild-type gametophores were cultured with 50 µg ml−1 zeocin and 10 µg ml−1 PI for 6 h, washed and cultured again with 10 µg ml−1 PI. Time-lapse observations were performed at 2 h intervals for 90 h. The red signals indicate PI fluorescence. The yellow arrows indicate reprogrammed cells with protrusion.
41477_2020_745_MOESM4_ESM.avi
Supplementary Video 2: PI fluorescence in leaves with dead cells of an intact gametophore during and after temporary zeocin treatment. Intact wild-type gametophores were cultured with 50 µg ml−1 zeocin and 10 µg ml−1 PI for 6 h, washed and cultured again with 10 µg ml−1 PI. Time-lapse observations were performed at 2 h intervals for 90 h. The red signals indicate PI fluorescence. The yellow and pink arrows indicate reprogrammed cells with protrusion and dead cells, respectively.
41477_2020_745_MOESM5_ESM.avi
Supplementary Video 3: Promoter activity of CYCD1;1 in leaves of an intact gametophore after temporary zeocin treatment. Intact gametophores of CYCD1;1pro:NGG line no. 263 were cultured in medium with 50 µg ml−1 zeocin for 6 h, washed and transferred to a glass-bottom dish. Time-lapse observations were performed at 2 h intervals for 98 h.
41477_2020_745_MOESM6_ESM.avi
Supplementary Video 4: Accumulation of CSP1-Citrine proteins in leaves of an intact gametophore after temporary zeocin treatment. Intact gametophores of nPpCSP1-Citrine-3ʹUTR no. 1 were cultured in medium with 50 µg ml−1 zeocin for 6 h, washed and transferred to a glass-bottom dish. Time-lapse observations were performed at 2 h intervals for 96 h.
41477_2020_745_MOESM7_ESM.avi
Supplementary Video 5: Promoter activity of STEMIN1 in leaves of an intact gametophore after temporary zeocin treatment. Intact gametophores of STEMIN1pro:NGG line no. 7 were cultured in medium with 50 µg ml−1 zeocin for 6 h, washed and transferred to a glass-bottom dish. Time-lapse observations were performed at 1.5 h intervals for 79 h.
41477_2020_745_MOESM8_ESM.avi
Supplementary Video 6: Promoter activity of STEMIN2 in leaves of an intact gametophore after temporary zeocin treatment. Intact gametophores of STEMIN2pro:NGG line no. 238 were cultured in medium with 50 µg ml−1 zeocin for 6 h, washed and transferred to a glass-bottom dish. Time-lapse observations were performed at 1.5 h intervals for 98 h.
41477_2020_745_MOESM9_ESM.avi
Supplementary Video 7: Promoter activity of STEMIN3 in leaves of an intact gametophore after temporary zeocin treatment. Intact gametophores of STEM3N1pro:NGG line no. 26 were cultured in medium with 50 µg ml−1 zeocin for 6 h, washed and transferred to a glass-bottom dish. Time-lapse observations were performed at 1.5 h intervals for 98 h.
Rights and permissions
About this article
Cite this article
Gu, N., Tamada, Y., Imai, A. et al. DNA damage triggers reprogramming of differentiated cells into stem cells in Physcomitrella. Nat. Plants 6, 1098–1105 (2020). https://doi.org/10.1038/s41477-020-0745-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-020-0745-9
This article is cited by
-
Mechanistic insights into DNA damage recognition and checkpoint control in plants
Nature Plants (2024)
-
Plant regeneration in the new era: from molecular mechanisms to biotechnology applications
Science China Life Sciences (2024)
-
Neural cell state shifts and fate loss in ageing and age-related diseases
Nature Reviews Neurology (2023)
-
Autopolyploidization affects transcript patterns and gene targeting frequencies in Physcomitrella
Plant Cell Reports (2022)
-
Fundamental mechanisms of the stem cell regulation in land plants: lesson from shoot apical cells in bryophytes
Plant Molecular Biology (2021)